The Mars-van Krevelen Mechanism: A Comprehensive Guide for Oxidation Catalysis in Biomedical Research
This article provides a detailed exploration of the Mars-van Krevelen (MvK) mechanism, a cornerstone concept in heterogeneous oxidation catalysis.
The Mars-van Krevelen Mechanism: A Comprehensive Guide for Oxidation Catalysis in Biomedical Research
Abstract
This article provides a detailed exploration of the Mars-van Krevelen (MvK) mechanism, a cornerstone concept in heterogeneous oxidation catalysis. Tailored for researchers, scientists, and drug development professionals, it covers foundational principles, key applications in chemical synthesis and environmental remediation, methodological considerations for catalyst design, and troubleshooting strategies for activity and selectivity. It further examines validation techniques, compares the MvK mechanism to alternative pathways like Langmuir-Hinshelwood, and discusses its emerging implications in biomimetic chemistry and therapeutic development.
Understanding the Mars-van Krevelen Mechanism: Core Principles and Catalytic Cycle
This whitepaper situates the discovery and foundational principles of the Mars-van Krevelen (MvK) mechanism within the broader, evolving thesis of its application in selective oxidation catalysis. Originally conceptualized for metal oxide catalysts, the core thesis posits that the MvK mechanism is not merely a surface redox cycle but a generalizable paradigm for understanding and engineering lattice oxygen participation in heterogeneous oxidation. This framework is now being rigorously tested and extended to complex materials, including doped oxides and metal-organic frameworks, with profound implications for sustainable chemical synthesis and energy-related transformations. The enduring relevance of the MvK thesis lies in its predictive power for catalyst design, where the kinetics of oxygen vacancy formation and replenishment dictate activity, selectivity, and stability.
Historical Discovery and Foundational Experiments
The mechanism was first explicitly formulated in the 1950s by Pieter Mars and Jan Hendrik (Dick) van Krevelen in their seminal work on the catalytic oxidation of hydrocarbons over solid oxide catalysts. Their key insight was that the oxygen incorporated into the oxidation product originated from the oxide catalyst lattice itself, not directly from gaseous oxygen. The gaseous O₂ subsequently re-oxidized the reduced catalyst, completing the cycle.
Foundational Experiment: Oxidation of Aromatic Hydrocarbons over V₂O₅ Catalysts Mars and van Krevelen studied the kinetics of naphthalene and benzene oxidation over V₂O₅ catalysts. By correlating reaction rates with the reducibility of various metal oxides and analyzing kinetic data, they proposed a two-step mechanism to explain the observed negative order in hydrocarbon and positive order in oxygen.
Detailed Methodology:
- Catalyst Preparation: High-purity V₂O₅ powder was pelletized and sieved to a specific particle size range (e.g., 100-200 µm) to minimize mass transfer limitations.
- Kinetic Rate Measurements: A fixed-bed flow reactor operating under differential conversion conditions (<15%) was used. The partial pressures of the hydrocarbon (e.g., naphthalene, ( P{HC} )) and oxygen (( P{O_2} )) were systematically varied.
- Isotopic Labeling (A Later Confirmatory Technique): Although not used in the original 1954 paper, subsequent key experiments employed ( ^{18}O_2 ) to confirm the lattice oxygen pathway.
- Protocol: Pre-treat the catalyst with ( ^{18}O2 ) to label the lattice oxygen. The gas phase is then switched to a mixture of hydrocarbon and natural ( ^{16}O2 ). The initial appearance of ( H2^{18}O ) or ( C^{18}O2 ) in the products, before any ( ^{18}O )-labeled oxygenated hydrocarbon, provides direct evidence for lattice oxygen insertion.
- Data Analysis: The rate equation was derived and fitted to the experimental data. The classic Mars-van Krevelen rate expression is: [ r = \frac{k1 k2 P{HC} P{O2}}{k1 P{HC} + k2 P{O2}} ] where ( k1 ) is the rate constant for hydrocarbon oxidation by lattice oxygen and ( k2 ) is the rate constant for catalyst re-oxidation by gas-phase O₂.
Table 1: Summary of Key Quantitative Findings from Foundational Studies
| Catalyst System | Reaction Studied | Observed Kinetic Orders | Key Evidence | Proposed Rate Constants (Arbitrary Units) |
|---|---|---|---|---|
| V₂O₅ | Naphthalene → Phthalic Anhydride | Hydrocarbon: ~0, Oxygen: Positive | Correlation of activity with oxide reducibility | ( k1 ): 5.2, ( k2 ): 3.1 |
| V₂O₅ / MoO₃ | Benzene → Maleic Anhydride | Hydrocarbon: Negative, Oxygen: Positive | Kinetic data fit to two-step model | ( k1 ): 8.7, ( k2 ): 1.8 |
| (^{18}O_2)-Labeled V₂O₅ | Propylene Oxidation | N/A | Detection of ( ^{18}O ) in initial CO₂/H₂O, not in acrolein | Confirms lattice oxygen pathway |
Core Mechanism and Modern Extensions
The canonical MvK mechanism involves a cyclic process of catalyst reduction and re-oxidation.
Experimental Protocols for Contemporary MvK Studies
Protocol A: Transient Isotopic Kinetic (TIK) Experiment
- Objective: To quantify the number and reactivity of lattice oxygen atoms participating in a reaction.
- Procedure:
- Pre-treatment: Catalyst is oxidized in pure ( ^{16}O2 ) at reaction temperature, then purged with inert gas.
- Switch to Labeled Feed: Introduce a pulse or steady flow of ( ^{18}O2 ) over the catalyst while monitoring the gas-phase composition via mass spectrometry.
- Data Analysis: The evolution of ( ^{18}O2 ), ( ^{16}O^{18}O ), and ( ^{16}O2 ) is tracked. The amount of ( ^{16}O ) exchanged quantifies the active lattice oxygen pool. The rate of exchange informs on the mobility of lattice oxygen.
Protocol B: Steady-State Isotopic Transient Kinetic Analysis (SSITKA)
- Objective: To measure the surface residence time and number of active intermediates under working reaction conditions.
- Procedure:
- Establish Steady State: Run the catalyst under a steady flow of reactants (e.g., ( C3H6 ), ( ^{16}O_2 ), He).
- Isotopic Step-Change: Abruptly switch the oxygen feed from ( ^{16}O2 ) to ( ^{18}O2 ) while maintaining all other conditions.
- Monitor Transients: Use MS to monitor the decay of ( ^{16}O )-containing products and the rise of ( ^{18}O )-containing products.
- Analysis: The mean surface residence time of the reacting oxygen intermediate is calculated from the transient curves, providing direct insight into the MvK cycle dynamics.
Modern Applications in Selective Oxidation: A Case Study
The selective oxidation of propylene to acrolein over bismuth molybdates (Bi-Mo-O) is a classic industrial application of the MvK mechanism. The catalyst's multilayer structure features specific sites for propylene activation (Bi³⁺) and oxygen insertion (Mo⁶⁺-O).
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Investigating the MvK Mechanism
| Reagent / Material | Function in MvK Research | Key Characteristics / Notes |
|---|---|---|
| (^{18})O₂ Isotope (≥97 atom %) | Tracer for lattice oxygen participation in TIK and SSITKA experiments. | Enables definitive proof of the MvK pathway by tracking labeled oxygen atoms. |
| High-Purity Metal Oxide Precursors (e.g., NH₄VO₃, (NH₄)₆Mo₇O₂₄, Bi(NO₃)₃) | Synthesis of well-defined, single-phase oxide catalysts (e.g., V₂O₅, Bi₂Mo₃O₁₂). | Phase purity is critical for attributing mechanistic findings to specific crystal structures. |
| Pulse Reaction Microreactor System | Allows transient kinetic experiments, including TIK and temperature-programmed techniques. | Coupled to MS or GC for rapid, time-resolved product analysis. |
| In Situ Raman Spectroscopy Cell | Monitors catalyst surface under reaction conditions; can identify surface oxide phases, peroxo species, and reduction states. | Provides molecular-level insight into the nature of the active oxygen species. |
| Operando X-ray Absorption (XAS) Cell | Tracks the oxidation state and local coordination environment of the metal cations in real-time during the redox cycle. | Directly measures the reduction (during hydrocarbon oxidation) and re-oxidation steps of the MvK cycle. |
| Density Functional Theory (DFT) Software (e.g., VASP, Quantum ESPRESSO) | Computes activation barriers for oxygen vacancy formation, oxygen migration, and C-H bond activation on model surfaces. | Used to validate and predict mechanistic steps at the atomic level, guiding catalyst design. |
This whitepaper provides an in-depth technical guide on the lattice oxygen participation pathway, a defining principle within the broader Mars-van Krevelen (MvK) mechanism for oxidation catalysis. It is designed for researchers and professionals seeking a rigorous, contemporary understanding of this critical heterogeneous catalytic process.
Core Principles of the Mars-van Krevelen Mechanism
The Mars-van Krevelen mechanism fundamentally departs from Langmuir-Hinshelwood or Eley-Rideal models by proposing that oxygen atoms from the bulk catalyst lattice, not just adsorbed molecular oxygen (O₂), directly participate in the oxidation of a reactant. The catalytic cycle comprises two interconnected half-cycles:
- Reduction Half-Cycle: A substrate molecule (e.g., CO, hydrocarbon) is oxidized by a surface lattice oxygen (O_lattice), creating an oxygen vacancy (□) and a partially reduced catalyst site.
- Re-oxidation Half-Cycle: The oxygen vacancy is replenished by the dissociation and incorporation of an oxidant (typically gaseous O₂), regenerating the O_lattice and closing the cycle.
The "lattice oxygen participation pathway" emphasizes the dynamic formation and healing of vacancies as the central kinetic and mechanistic feature.
Quantitative Metrics and Descriptors
Key quantitative parameters for evaluating and comparing catalysts operating via the MvK mechanism are summarized below.
Table 1: Key Quantitative Descriptors for Lattice Oxygen Pathways
| Descriptor | Symbol/Unit | Definition & Experimental Method | Typical Range (Exemplar Systems) |
|---|---|---|---|
| Oxygen Storage Capacity | OSC (μmol O₂/g) | Quantity of reactive lattice oxygen available for participation. Measured via H₂-TPR or CO-pulse chemisorption. | 100 - 500 μmol/g (CeO₂-based) |
| Oxygen Vacancy Formation Energy | E_form (eV) | Energy required to remove a surface lattice oxygen atom, creating a vacancy. Calculated via DFT. | 1.5 - 3.5 eV (Transition Metal Oxides) |
| Surface Lattice Oxygen Reactivity | TOF_O (s⁻¹) | Turnover frequency based on active surface O_lattice sites. Derived from kinetic isotopic labeling. | 10⁻³ - 10¹ s⁻¹ (at 300°C) |
| ¹⁸O₂ Isotope Exchange Rate | R_exchange (mol O₂/m²·s) | Rate of exchange between gaseous ¹⁸O₂ and bulk ¹⁶O_lattice. Measured via SSITKA-MS. | Varies by oxide reducibility |
| Activation Energy for Re-oxidation | E_a,ox (kJ/mol) | Barrier for dissociative O₂ adsorption and vacancy filling. Determined from kinetic studies. | 20 - 100 kJ/mol |
Experimental Protocols for Probing Lattice Oxygen Pathways
Protocol 3.1: Steady-State Isotopic Transient Kinetic Analysis (SSITKA) with ¹⁸O₂
Objective: To decouple the reduction and re-oxidation half-cycles and quantify the participation rate of bulk lattice oxygen. Materials: Fixed-bed microreactor, Mass Spectrometer (MS), ¹⁶O₂/¹⁸O₂ gas switching system, catalyst sample. Procedure:
- Establish steady-state catalytic oxidation (e.g., CO oxidation) under a flow of 2% CO in ¹⁶O₂/He at reaction temperature (e.g., 300°C).
- At time t=0, perform a rapid switch from the ¹⁶O₂ feed to an isotopically identical ¹⁸O₂ feed without perturbing concentration, flow, or pressure.
- Monitor the transient response of reaction products (e.g., C¹⁶O₂, C¹⁶O¹⁸O, C¹⁸O₂) and the ¹⁸O₂ feed via MS.
- Analyze the decay profile of C¹⁶O₂ and the rise profiles of the mixed isotopes. The time delay and distribution of ¹⁸O in the product directly quantify the number and lifetime of active oxygen pools participating in the reaction.
Protocol 3.2: In Situ Raman Spectroscopy during Catalytic Reaction
Objective: To identify the nature of surface oxygen species and monitor dynamic vacancy formation in real-time. Materials: In situ Raman cell reactor, laser excitation source (e.g., 532 nm), spectrometer, temperature/gas control. Procedure:
- Load catalyst as a thin wafer into the in situ cell. Pre-treat in O₂ flow at 500°C, then cool to reaction temperature in inert gas.
- Begin collecting reference spectra under inert atmosphere.
- Introduce reactant mixture (e.g., propene + He). Continuously collect Raman spectra (e.g., every 30 seconds).
- Observe changes in key vibrational bands: disappearance/intensity reduction of metal-O lattice bands (e.g., ~600 cm⁻¹ for perovskites), appearance of bands associated with reduced phases or intermediate surface species.
- Switch back to O₂ flow to observe the recovery (re-oxidation) of the lattice bands, confirming the reversible vacancy formation.
Essential Research Reagent Solutions & Materials
Table 2: The Scientist's Toolkit for MvK Pathway Studies
| Item | Function in Research |
|---|---|
| ¹⁸O₂ Isotope (97%+ enrichment) | Tracer gas for SSITKA and isotope exchange experiments to track oxygen atom pathways. |
| Cerium-Zirconium Mixed Oxide (CeₓZr₁₋ₓO₂) | Model redox oxide with high, tunable OSC and vacancy mobility. |
| Perovskite Catalysts (e.g., La₀.₈Sr₀.₂CoO₃₋δ) | Model systems for studying non-stoichiometry (δ) and the role of cation substitution in O_lattice reactivity. |
| Probe Molecules (CO, H₂, C₃H₆) | Standard reductants for Temperature-Programmed Reduction (TPR) and pulse experiments to measure OSC and reactivity. |
| In Situ/Operando Cells | Specialty reactors (IR, Raman, XRD) allowing spectroscopic characterization under working catalytic conditions. |
Visualization of Pathways and Workflows
Diagram 1: The Mars-van Krevelen Catalytic Cycle
Diagram 2: SSITKA with ¹⁸O₂ Experimental Workflow
Within the broader thesis of Mars-van Krevelen (MvK) mechanism research for catalytic oxidation, a fundamental challenge lies in its unambiguous experimental discrimination from traditional Langmuir-Hinshelwood (LH) and Eley-Rideal (ER) adsorption-based pathways. This guide provides a technical framework for their distinction.
Foundational Mechanistic Pathways
The core distinction lies in the role of the catalyst's lattice oxygen.
Title: Core Mechanistic Pathways for Catalytic Oxidation
Quantitative Discriminatory Criteria
Experimental data must be evaluated against key comparative metrics.
Table 1: Discriminatory Characteristics Between Mechanisms
| Characteristic | Mars-van Krevelen (MvK) | Langmuir-Hinshelwood (LH) | Eley-Rideal (ER) |
|---|---|---|---|
| Oxygen Source | Catalyst lattice oxygen | Chemisorbed molecular oxygen | Chemisorbed or gas-phase oxygen |
| Rate Dependence on O₂ | Weak or zero-order at moderate pressures | Often first-order at low pressure | Variable; can be zero-order for adsorbed oxidant |
| Reductant Order | Often first-order | Can be negative at high coverage | First-order |
| Isotope Exchange (¹⁸O₂) | Rapid exchange with lattice oxygen even without reductant | Exchange only via adsorption/desorption on sites | Limited exchange, requires reactant |
| Kinetic Isotope Effect (KIE) | Often shows a significant KIE using ¹⁸O-lattice | Typically small KIE from O₂ activation | Small or no KIE from oxidant |
| Catalyst Oxidation State | Cycles during reaction (e.g., Ce⁴⁺/Ce³⁺, V⁵⁺/V⁴⁺) | Remains relatively constant | Remains relatively constant |
| Transient Response to O₂ Cut-off | Reaction continues until lattice O depleted; slow recovery | Stops abruptly; fast recovery | Stops abruptly; fast recovery |
Table 2: Summary of Key Quantitative Evidence from Recent Studies (2020-2023)
| Catalyst System | Probed Reaction | Key Evidence for MvK | Experimental Technique | Ref. |
|---|---|---|---|---|
| Co₃O₄ Nanocrystals | CO Oxidation | Rate independent of O₂ pressure; high ¹⁸O₂ exchange rate | Temporal Analysis of Products (TAP), SSITKA | [1] |
| CeO₂-Based Single-Atom | CH₄ Oxidation | Direct observation of lattice O involvement via in situ Raman; KIE = 1.8 | In situ Raman, Isotopic Labelling | [2] |
| V₂O₅/WO₃-TiO₂ | NH₃-SCR (NOx reduction) | Rate proportional to V⁵⁺=O site concentration; ¹⁶O-¹⁸O scrambling | Operando XAS, FTIR, Isotope Switching | [3] |
Experimental Protocols for Distinction
Protocol 1: Transient Isotope Labelling (Steady-State Isotopic Transient Kinetic Analysis - SSITKA)
- Objective: Trace the fate of lattice vs. gaseous oxygen.
- Method:
- Establish steady-state reaction (e.g., CO + ¹⁶O₂ → CO₂) in a plug-flow reactor.
- Switch the feed abruptly from ¹⁶O₂ to isotopically labelled ¹⁸O₂ while maintaining total flow and concentration.
- Monitor product evolution (e.g., C¹⁶O₂, C¹⁶O¹⁸O, C¹⁸O₂) in real-time using mass spectrometry (MS).
- Interpretation: Immediate appearance of C¹⁸O₂ suggests an ER/LH pathway. A gradual transition from C¹⁶O₂ to C¹⁸O₂, with mixed C¹⁶O¹⁸O, indicates lattice oxygen participation (MvK), as the lattice ¹⁶O is gradually replaced by ¹⁸O.
Protocol 2: Oxygen Isotope Exchange (OIE) Experiment
- Objective: Measure catalyst's inherent lattice oxygen mobility.
- Method:
- Pre-treat catalyst in ¹⁶O₂.
- Switch to isotopically labelled ¹⁸O₂ gas stream in the absence of the reductant.
- Monitor the evolution of ¹⁶O¹⁸O (mass 34) and depletion of ¹⁸O₂ (mass 36) via MS.
- Interpretation: High rates of ¹⁶O¹⁸O formation indicate facile lattice oxygen exchange, a prerequisite for the MvK mechanism. Adsorption-based mechanisms show minimal exchange without a reducing agent.
Protocol 3: Transient Pulse Response (TAP Reactor)
- Objective: Probe elementary steps and kinetic constants.
- Method:
- Place catalyst in a vacuum microreactor.
- Inject ultra-short, alternate pulses of reductant (e.g., CO) and oxidant (O₂).
- Analyze the temporal distribution of product pulses at the reactor exit with MS.
- Interpretation: If a CO pulse yields CO₂ without a concurrent O₂ pulse, it directly proves oxidation by lattice oxygen (MvK). Sequential pulsing can then measure the rate constant for lattice oxygen regeneration.
Mechanistic Decision Workflow
Title: Decision Workflow for Mechanism Discrimination
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Mechanistic Studies
| Item | Function & Relevance | Example/Specification |
|---|---|---|
| Isotopically Labelled Gases | To trace oxygen origin and pathway via SSITKA and OIE. | ¹⁸O₂ (97%+ enrichment), ¹³C¹⁶O, D₂ (for C-H bond KIE). |
| Model Oxide Catalysts | Well-defined surfaces for fundamental studies. | Single-crystal metal oxides (e.g., TiO₂(110), CeO₂(111)), supported vanadia, Co₃O₄ nanocrystals. |
| Mass Spectrometer (MS) | Real-time, quantitative tracking of isotopes and reaction products. | High-sensitivity, low-dead-volume quadrupole MS with fast response. |
| TAP Reactor System | For probing intrinsic kinetics and elementary steps via pulse experiments. | Ultra-high vacuum compatible, with piezoelectric pulse valves. |
| In Situ/Operando Cells | To observe catalyst state under reaction conditions. | High-temperature/pressure cells for Raman, FTIR, or XAS. |
| Calibrated Flow Controllers | For precise control of gas composition and transient switches. | MFCs with fast response time and high accuracy for dilute streams. |
| Reference Reductants | Standard probes for oxidation activity. | High-purity CO, CH₄, C₃H₆, H₂. |
This technical guide examines the defining properties of common redox metal oxides (V, Mo, Ce, Fe) within the critical framework of Mars-van Krevelen (MvK) mechanism research. The MvK mechanism is a cornerstone theory for heterogeneous oxidation catalysis, wherein the catalyst lattice oxygen actively participates in the reaction. The oxidant re-oxidizes the reduced catalyst, completing the catalytic cycle. The efficacy of this cycle is intrinsically governed by the physicochemical properties of the metal oxide. This whitepaper synthesizes current data and methodologies to elucidate how the properties of these specific oxides dictate their performance as MvK catalysts, providing a foundation for advanced research in catalytic oxidation and related fields like selective oxidation in pharmaceutical intermediate synthesis.
Core Properties of Redox Metal Oxides
The catalytic activity and selectivity in the MvK cycle are determined by several interlinked properties: redox potential, oxygen mobility, lattice structure, and acid-base character. The table below summarizes quantitative and qualitative data for the featured oxides.
Table 1: Comparative Properties of Selected Redox Metal Oxides in MvK Catalysis
| Property / Oxide | Vanadium Oxides (e.g., V₂O₅) | Molybdenum Oxides (e.g., MoO₃) | Cerium Oxide (CeO₂, Ceria) | Iron Oxides (e.g., α-Fe₂O₃, Fe₃O₄) |
|---|---|---|---|---|
| Common MvK Applications | Selective oxidation of hydrocarbons (e.g., o-xylene to phthalic anhydride), SO₂ oxidation, NOx reduction. | Selective oxidation and ammoxidation of propylene to acrolein/acrylonitrile, methanol to formaldehyde. | Three-way catalysts (TWCs), CO oxidation, water-gas shift, soot combustion, VOC removal. | Fischer-Tropsch synthesis, dehydrogenation of ethylbenzene to styrene, water-gas shift, Fenton-like reactions. |
| Redox Couples | V⁵⁺/V⁴⁺, V⁴⁺/V³⁺ | Mo⁶⁺/Mo⁵⁺, Mo⁵⁺/Mo⁴⁺ | Ce⁴⁺/Ce³⁺ | Fe³⁺/Fe²⁺ |
| Oxygen Mobility | Moderate. Layer structure allows for reversible oxygen removal. | Low to Moderate. Requires specific crystalline faces for activity. | Very High. High concentration of oxygen vacancies and superior oxygen storage capacity (OSC). | Moderate to High. Depends on phase and dopants; facile Fe redox cycling. |
| Key Structural Feature | Layered, distorted trigonal bipyramidal or octahedral coordination. | Layered structure with corner- and edge-sharing MoO₆ octahedra. | Fluorite structure; facile formation of oxygen vacancies. | Various phases (Hematite, Magnetite); stable under diverse conditions. |
| Acid-Base Character | Strong acidic sites (Brønsted and Lewis). | Predominantly acidic sites. | Amphoteric (oxygen vacancies act as Lewis acid, surface OH as Brønsted base). | Typically basic, but can be tuned with dopants. |
| Typical Dopants/ Promoters | P, Mo, Sb, Na, K (modify acidity/redox). | V, Te, Sb, Bi (for multicomponent catalysts like MoVTeNbO). | Zr, La, Pr, Gd (enhance thermal stability and OSC). | Cr, Mn, Ce, Zr (enhance stability and redox properties). |
| Quantitative Metric (OSC) | ~ 50 - 200 μmol O₂/g (varies with support/promoters) | Low intrinsic OSC | > 400 μmol O₂/g (for pure nano-ceria, can exceed 600 with doping) | ~ 100 - 300 μmol O₂/g (highly dependent on preparation) |
Experimental Protocols for MvK Mechanism Investigation
Protocol 1: Isotopic Oxygen Exchange (IOE) and Temporal Analysis of Products (TAP)
Objective: To directly prove lattice oxygen participation and quantify its reactivity. Methodology:
- Pretreatment: The catalyst (~100 mg) is loaded in a microreactor and pre-oxidized in a stream of 5% O₂/He at 500°C for 1 hour, then purged with He.
- ¹⁸O₂ Isotopic Pulse: The reactor is switched to a pulse mode (e.g., using a TAP reactor system). A calibrated pulse of ¹⁸O₂ (isotopically labeled oxygen) is introduced.
- Mass Spectrometry Analysis: The effluent is monitored by a high-speed mass spectrometer (MS). The evolution of gaseous ¹⁶O₂ (m/z=32), ¹⁶O¹⁸O (m/z=34), and ¹⁸O₂ (m/z=36) is tracked in real-time.
- Data Interpretation: The rapid appearance of ¹⁶O¹⁸O and ¹⁶O₂ indicates exchange between gaseous ¹⁸O₂ and lattice ¹⁶O, confirming mobile lattice oxygen. The kinetics of exchange inform on oxygen diffusion rates.
Protocol 2: Steady-State Isotopic Transient Kinetic Analysis (SSITKA)
Objective: To measure the surface residence time and inventory of active intermediates during steady-state reaction conditions. Methodology:
- Achieve Steady State: A steady-state reaction flow (e.g., 1% CO + 1% O₂ in He) is established over the catalyst at reaction temperature. Conversion and rates are stabilized.
- Isotopic Switch: The feed is abruptly switched to an isotopically labeled, otherwise identical stream (e.g., switching from ¹²C¹⁶O + ¹⁶O₂ to ¹³C¹⁸O + ¹⁸O₂) while maintaining total flow and composition.
- Transient Response Monitoring: The MS monitors the decay of the unlabeled product (e.g., ¹²C¹⁶O₂) and the rise of the labeled product (e.g., ¹³C¹⁸O₂).
- Analysis: The mean surface residence time (τ) of the reacting carbon/intermediate is calculated from the transient curves. A long τ for oxygen in the product relative to the feed switch time provides direct evidence for a pool of active lattice oxygen (the MvK pool).
Protocol 3: In Situ Raman Spectroscopy under Reaction Conditions
Objective: To identify the specific surface oxide phases and defect states (like V=O, Mo=O, or oxygen vacancies) during reaction. Methodology:
- Cell Setup: Catalyst powder is placed in a high-temperature in situ Raman cell with optical windows, capable of gas flow and temperature control.
- Spectra Collection: Spectra are first collected under pre-oxidation conditions (in O₂). The gas feed is then switched to the reactant mixture (e.g., propane + O₂ or just propane).
- Dynamic Monitoring: Raman spectra are collected continuously (e.g., every 30 seconds). Changes in key vibrational bands (e.g., ~1030 cm⁻¹ for V=O in V₂O₅, ~460 cm⁻¹ for F₂g mode of CeO₂ indicating vacancy formation) are tracked.
- Correlation: The decrease in metal-oxygen bond Raman intensity coupled with the appearance of reduced phase bands directly correlates lattice oxygen consumption with reactant oxidation.
Visualization of the Mars-van Krevelen Mechanism
The Scientist's Toolkit: Key Research Reagent Solutions & Materials
Table 2: Essential Materials for Redox Metal Oxide & MvK Mechanism Studies
| Item / Reagent | Function & Rationale |
|---|---|
| High-Purity Metal Oxide Precursors (e.g., NH₄VO₃, (NH₄)₆Mo₇O₂₄·4H₂O, Ce(NO₃)₃·6H₂O, Fe(NO₃)₃·9H₂O) | Starting materials for catalyst synthesis via methods like precipitation, sol-gel, or impregnation. High purity minimizes confounding impurities that alter redox or acidic properties. |
| Isotopically Labeled Gases (¹⁸O₂ (97%+), ¹³CO, D₂) | Critical for mechanistic probes (IOE, SSITKA) to trace the pathway of oxygen and other atoms, providing unambiguous evidence for the MvK pathway. |
| Temperature-Programmed Reaction (TPRx) Setup | A system comprising mass flow controllers, a tubular reactor in a programmable furnace, and an online MS or GC. Used for redox property profiling (H₂-TPR, O₂-TPO) and testing reaction pathways. |
| In Situ/Operando Cell (for Raman, DRIFTS, or XRD) | A reaction cell with gas flow and heating, compatible with spectroscopic beamlines. Allows observation of catalyst structure and surface species under actual reaction conditions, bridging the pressure gap. |
| Chemisorption Analyzer | For quantifying surface area (BET), acid site density/dstrength (via NH₃-TPD), and metal dispersion. Correlates physical structure with catalytic performance. |
| Redox Buffers & Dopant Salts (e.g., ZrOCl₂, La(NO₃)₃, Sb₂O₃) | Used to modify the host oxide's properties (e.g., doping CeO₂ with Zr to enhance thermal stability and oxygen mobility). |
| Model Reductant/Oxidant Probes (e.g., CO, CH₄, C₃H₆, H₂O₂) | Well-characterized molecules used to test specific aspects of catalyst functionality (e.g., CO oxidation probes low-temperature redox activity). |
| High-Surface-Area Supports (e.g., TiO₂ (P25), γ-Al₂O₃, SiO₂, Carbon) | Used to disperse active oxide phases, increase accessible surface area, and potentially induce strong metal-support interactions (SMSI) that modify redox properties. |
Thesis Context: This whitepaper provides an in-depth technical examination of the thermodynamic and kinetic principles governing lattice oxygen mobility and catalyst regeneration, framed within the broader research on the Mars-van Krevelen (MvK) mechanism for catalytic oxidation reactions.
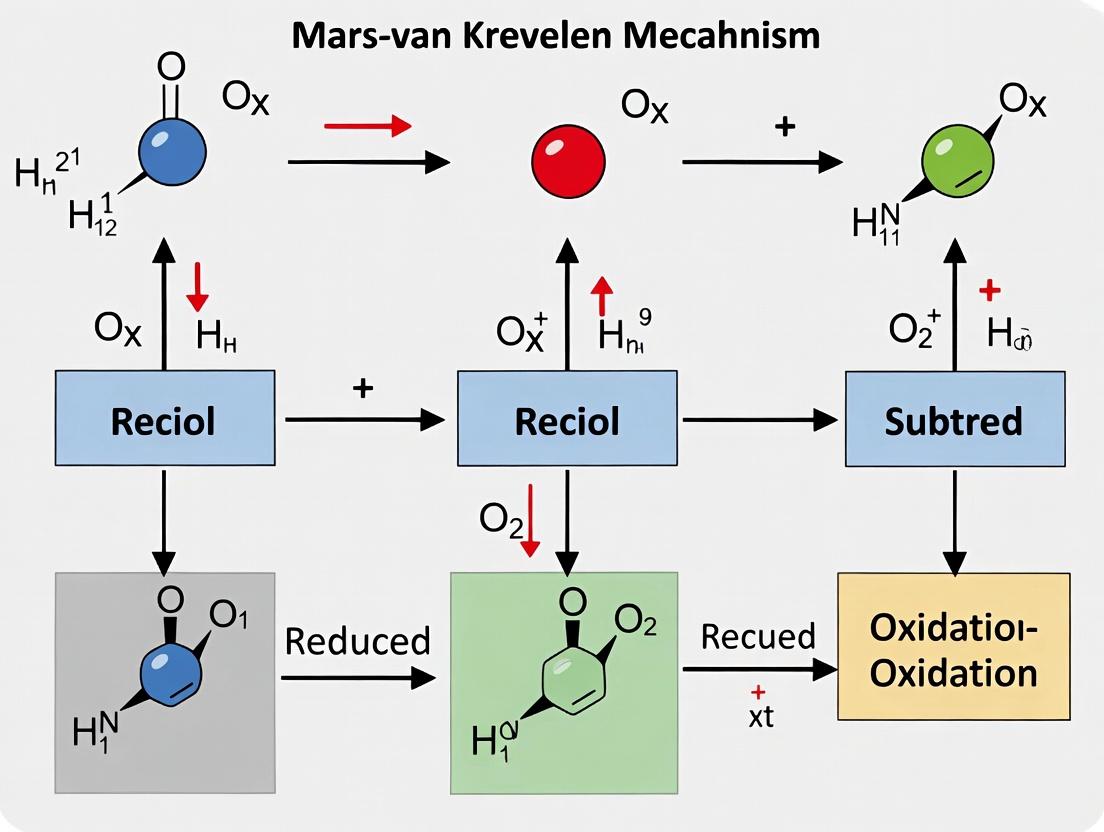
The Mars-van Krevelen mechanism is a cornerstone of heterogeneous oxidation catalysis. It describes a process where a substrate is oxidized by lattice oxygen from a metal oxide catalyst, leaving an oxygen vacancy. The catalyst is subsequently regenerated by a gaseous oxidant (e.g., O₂), which replenishes the lattice oxygen. The efficiency of this catalytic cycle is fundamentally controlled by two intertwined factors: the thermodynamic driving force for oxygen vacancy formation and the kinetic mobility of lattice oxygen ions.
Core Principles: Thermodynamics vs. Kinetics
Thermodynamic Drivers
Thermodynamics dictates the feasibility of lattice oxygen extraction. The key parameter is the oxygen vacancy formation energy (Ef), which is the energy required to remove a lattice oxygen atom, forming a vacancy and releasing a gaseous O₂ molecule. A lower Ef generally indicates a more easily reducible oxide and a higher thermodynamic driving force for the oxidation step.
Kinetic Drivers
Kinetics governs the rate at which oxygen moves through the catalyst bulk and surface. The oxygen diffusion coefficient (D) and the surface exchange coefficient (k) are critical parameters. High mobility ensures rapid replenishment of active surface oxygen from the bulk, preventing catalyst deactivation.
Quantitative Data on Representative Catalytic Oxides
The following table summarizes key thermodynamic and kinetic parameters for oxides commonly studied within MvK research, such as in selective oxidation and total combustion.
Table 1: Thermodynamic and Kinetic Parameters for Selected Metal Oxides in MvK Catalysis
| Oxide Catalyst | Primary Application (MvK Context) | Approx. Oxygen Vacancy Formation Energy (E_f in eV) | Oxygen Diffusion Coefficient (D at 600°C in cm²/s) | Surface Exchange Coefficient (k at 600°C in cm/s) |
|---|---|---|---|---|
| CeO₂ (Ceria) | CO Oxidation, VOC Combustion | 2.0 - 3.0 (highly dependent on doping) | ~10⁻¹⁰ to 10⁻⁸ | ~10⁻⁷ to 10⁻⁵ |
| V₂O₅ | Selective Oxidation of Hydrocarbons (e.g., o-xylene to phthalic anhydride) | ~2.5 - 3.5 | ~10⁻¹⁴ | ~10⁻¹⁰ |
| MoO₃ | Selective Oxidation (e.g., methanol to formaldehyde) | ~3.0 - 4.0 | ~10⁻¹⁶ | ~10⁻¹² |
| Co₃O₄ | N₂O Decomposition, Low-Temp CO Oxidation | ~2.0 - 2.5 | ~10⁻¹¹ | ~10⁻⁸ |
| Perovskite (LaCoO₃) | Soot Combustion, Methane Oxidation | Tunable via A/B site doping (1.5 - 4.0) | Tunable (10⁻¹⁵ to 10⁻¹⁰) | Tunable (10⁻¹² to 10⁻⁷) |
Experimental Protocols for Probing Oxygen Mobility and Regeneration
Isotopic Oxygen Exchange (IOE) Experiment
This is the definitive method for measuring surface exchange (k) and bulk diffusion (D) coefficients.
Detailed Protocol:
- Preparation: Place 100-200 mg of catalyst powder in a quartz tubular microreactor. Connect to a closed gas-circulation system or a pulse reactor system coupled to a mass spectrometer (MS).
- Pre-treatment: Heat the sample to the desired temperature (e.g., 500-800°C) under flowing inert gas (He) to clean the surface. Then expose to natural O₂ (¹⁶O₂) until saturation.
- Isotopic Switch: Rapidly switch the gas feed to a mixture of ²⁸O₂ (or ¹⁸O₂) in He (e.g., 2% O₂ in He). The total pressure is maintained at 1 atm.
- MS Monitoring: Use the MS to continuously monitor the concentrations of different oxygen isotopologues in the gas phase: ³²O₂ (¹⁶O-¹⁶O), ³⁴O₂ (¹⁶O-¹⁸O), and ³⁶O₂ (¹⁸O-¹⁸O).
- Data Analysis: Model the temporal evolution of the isotopologue concentrations using the Fick's second law and appropriate boundary conditions to extract the chemical diffusion coefficient (D) and the surface exchange coefficient (k).
Temperature-Programmed Reduction (TPR) and Reoxidation (TPO)
Used to assess reducibility (related to E_f) and regeneration capacity.
Detailed Protocol (TPR):
- Loading: Load 50 mg of catalyst into a U-shaped quartz reactor.
- Gas Flow: Pass a 5% H₂/Ar reducing gas mixture over the sample at a constant flow rate (e.g., 30 mL/min).
- Temperature Ramp: Heat the reactor from room temperature to 900°C at a linear rate (e.g., 10°C/min).
- Detection: Measure hydrogen consumption using a thermal conductivity detector (TCD). Peaks indicate reduction events at specific temperatures, with lower temperatures suggesting easier lattice oxygen removal.
Detailed Protocol (TPO - for Regeneration Study):
- Pre-reduction: First, partially reduce the catalyst using a controlled pulse of H₂ or under reaction conditions.
- Cooling & Purging: Cool to a standard temperature (e.g., 100°C) under inert gas.
- Reoxidation: Switch to a 2% O₂/He flow and perform a temperature ramp (e.g., 10°C/min) up to 800°C.
- Detection: Monitor O₂ consumption with a TCD or MS. The temperature and quantity of O₂ consumed reveal the kinetics and completeness of lattice oxygen regeneration.
In Situ Raman Spectroscopy for Detecting Oxygen Species
Detailed Protocol:
- Cell Setup: Place catalyst in a high-temperature, controlled-atmosphere in situ Raman cell.
- Reaction Conditions: Flow reactant mixture (e.g., propane + He) over the catalyst while heating to reaction temperature (e.g., 500°C).
- Spectral Acquisition: Acquire Raman spectra continuously or at intervals. Monitor the decrease in intensity of bands associated with M-O-M lattice vibrations.
- Regeneration Phase: Switch flow to O₂/He. Observe the recovery of lattice oxygen bands and the appearance/disappearance of superoxide (O₂⁻) or peroxide (O₂²⁻) intermediate species, providing direct evidence of the oxygen regeneration pathway.
Visualization of Concepts and Workflows
Diagram 1: The Mars-van Krevelen Catalytic Cycle (59 chars)
Diagram 2: Oxygen Regeneration Pathway on Surface (61 chars)
Diagram 3: Isotopic Oxygen Exchange Experiment Workflow (68 chars)
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for MvK Oxygen Mobility Studies
| Reagent/Material | Function in Research | Key Consideration |
|---|---|---|
| Isotopically Labeled Oxygen (¹⁸O₂) | Core reagent for IOE experiments to trace oxygen diffusion and exchange pathways. | High isotopic purity (>95%) is critical for accurate MS data interpretation. |
| High-Purity Metal Oxide Powders (CeO₂, V₂O₅, Perovskites) | Model catalyst systems for fundamental studies of structure-activity-mobility relationships. | Precise control of synthesis method (e.g., sol-gel, combustion) to ensure reproducible morphology and defect density. |
| Controlled Atmosphere Cells (for XRD, Raman, FTIR) | Enable in situ or operando characterization of catalyst structure and surface species under reaction conditions. | Must withstand high temperatures (up to 1000°C) and have chemically inert windows (e.g., quartz, CaF₂). |
| Pulse Reactor System with MS/TCD | For performing transient kinetic experiments like IOE, TPR, and TPO to measure kinetic parameters. | Requires fast-response valves and a directly coupled, high-sensitivity mass spectrometer for real-time gas analysis. |
| Temperature-Programmed Desorption (TPD) Standards | Used to calibrate and quantify acid/base sites or oxygen desorption profiles that compete with MvK pathways. | Common standards include NH₃ for acidity and CO₂ for basicity. |
| Dopant Precursor Salts (e.g., Gd(NO₃)₃ for CeO₂) | To synthesize doped oxides, a primary strategy for tuning oxygen vacancy formation energy (E_f) and mobility. | Precursor choice affects doping efficiency and final material homogeneity. Nitrates and acetates are common. |
Implementing MvK Catalysis: Techniques, Synthesis, and Biomedical Applications
This technical guide details the synthesis and characterization of metal oxide catalysts active in oxidation reactions proceeding via the Mars-van Krevelen (MvK) mechanism. Within the broader thesis of MvK research, the preparation of materials with labile lattice oxygen is paramount. This protocol focuses on creating and validating catalysts where the lattice oxygen participates directly in the oxidation cycle, a cornerstone for applications in chemical synthesis and environmental catalysis.
Core Principles of MvK-Active Oxides
The MvK mechanism involves the oxidation of a substrate by lattice oxygen from the catalyst, creating an oxygen vacancy. The catalyst is subsequently re-oxidized by a gaseous oxidant (e.g., O₂), completing the cycle. Active materials typically feature:
- Reducible Metal Cations: (e.g., V⁵⁺/⁴⁺, Mo⁶⁺/⁵⁺, Ce⁴⁺/³⁺) capable of multiple oxidation states.
- Labile Lattice Oxygen: Weaker metal-oxygen bonds facilitating oxygen transfer.
- Structural Defects: Which serve as active sites for vacancy formation and migration.
Synthesis Protocols
Citric Acid Sol-Gel Synthesis (for Mixed Oxides, e.g., MoVTeNb-Ox)
This method promotes atomic-level mixing of precursors, essential for forming the complex M1 and M2 phases active in propane oxidation.
Materials:
- Ammonium metavanadate (NH₄VO₃)
- Ammonium heptamolybdate tetrahydrate ((NH₄)₆Mo₇O₂₄·4H₂O)
- Telluric acid (H₆TeO₆) or Te metal dissolved in H₂O₂/HNO₃
- Niobium(V) oxalate ammonium complex (C₁₀H₂₅N₃O₁₉Nb)
- Citric acid monohydrate (C₆H₈O₇·H₂O)
- Deionized water
Procedure:
- Precursor Solutions: Separately dissolve each metal precursor in warm deionized water. For Nb, use the oxalate complex solution directly. For Te, use a solution of telluric acid.
- Combination: Sequentially combine the Mo, V, and Te solutions under vigorous stirring at 60°C.
- Complexation: Add a 1.5x molar excess (relative to total metals) of citric acid to the mixed solution.
- Nb Addition: Slowly add the niobium oxalate solution to the stirring mixture.
- Gelation: Evaporate water at 80-90°C with continuous stirring until a viscous gel forms.
- Drying: Dry the gel overnight at 120°C in an oven to obtain a solid xerogel.
- Calcination: Place the xerogel in a quartz boat. Heat in a tubular furnace under a controlled atmosphere (e.g., N₂ or air) according to the following program:
- Ramp from RT to 350°C at 2°C/min, hold for 1 hour.
- Ramp to 600°C at 5°C/min, hold for 2 hours.
- Cool naturally to room temperature.
- Post-Treatment: Grind the resulting solid to a fine powder (<100 µm).
Hydrothermal Synthesis (for V₂O₅ and MoO₃-based Catalysts)
Procedure:
- Dissolve vanadyl sulfate (VOSO₄) or ammonium molybdate in water.
- Adjust pH to 9-10 with ammonium hydroxide to form a precipitate/solution.
- Transfer the mixture to a Teflon-lined stainless-steel autoclave (fill factor: 70%).
- Heat at 180°C for 24-48 hours.
- Cool naturally, filter, wash with water and ethanol, and dry at 100°C.
- Calcine in static air at 450°C for 4 hours.
Characterization Techniques & Quantitative Data
Characterization validates the presence of MvK-active features. Data is summarized from recent literature searches.
Table 1: Key Characterization Data for Model MvK Catalysts
| Catalyst System | Synthesis Method | BET Surface Area (m²/g) | Primary Phase (XRD) | Reducibility (H₂-TPR Peak Temp., °C) | Labile Oxygen Quantified (O₂-TPD or CO-TPR, μmol O/g) | Reference Activity (e.g., Propane Conversion % @ 400°C) |
|---|---|---|---|---|---|---|
| MoVTeNbOx (M1 phase) | Sol-Gel (Citric Acid) | 15 - 25 | Orthorhombic M1 | 520, 630 | 250 - 350 | 45 - 65% |
| V₂O₅/TiO₂ (10 wt%) | Wet Impregnation | 80 - 100 | Monolayer VOx on TiO₂ (Anatase) | 480 | 120 - 180 | 25% (Propane) |
| CeO₂-ZrO₂ (CZO) | Co-precipitation | 60 - 80 | Cubic Fluorite | 450, 580 | >500 | 80% (CO Oxidation @ 350°C) |
| α-MoO₃ Nanoplates | Hydrothermal | 5 - 10 | Orthorhombic α-MoO₃ | 680 | 80 - 120 | 15% (Methanol) |
Table 2: XPS Surface Analysis of Selected Catalysts
| Catalyst | V⁵⁺/(V⁴⁺+V⁵⁺) Ratio | Olatt / Oads Ratio | Ce³⁺/(Ce³⁺+Ce⁴⁺) (for CZO) |
|---|---|---|---|
| MoVTeNbOx (Active) | 0.65 - 0.75 | 1.5 - 2.5 | - |
| V₂O₅/TiO₂ | 0.85 - 0.95 | 0.8 - 1.2 | - |
| Ce₀.₅Zr₀.₅O₂ | - | 3.0 - 4.0 | 0.25 - 0.30 |
Experimental Protocol for Key Characterization: Temperature-Programmed Reduction (H₂-TPR)
Objective: To quantify reducibility and identify distinct oxygen species.
Materials: 50 mg sieved catalyst (250-355 µm), 5% H₂/Ar gas, thermal conductivity detector (TCD).
Procedure:
- Pre-treatment: Load catalyst into a U-shaped quartz reactor. Heat to 300°C at 10°C/min under Ar flow (30 mL/min). Hold for 1 hour to remove adsorbed species. Cool to 50°C.
- Baseline Stabilization: Switch to 5% H₂/Ar (30 mL/min). Allow TCD signal to stabilize.
- Reduction Run: Heat the reactor from 50°C to 900°C at a ramp rate of 10°C/min.
- Data Analysis: Record the TCD signal versus temperature. Calibrate peak areas using a known mass of pure CuO standard. Report reduction peak temperatures and total H₂ consumption (μmol/g).
Visualization of Concepts and Workflows
Title: The Mars-van Krevelen Catalytic Cycle
Title: Sol-Gel Synthesis and Validation Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for MvK Catalyst Research
| Item/Chemical | Primary Function in Synthesis/Testing | Notes for Handling & Selection |
|---|---|---|
| Ammonium Metavanadate (NH₄VO₃) | V precursor. Provides vanadium in soluble form for mixed oxides. | Dissolve in warm water (<60°C) or oxalic acid solution to prevent decomposition. |
| Niobium Oxalate Complex (C₁₀H₂₅N₃O₁₉Nb) | Nb precursor. Stable, water-soluble source of Nb⁵⁺. | Critical for MoVTeNbOx. Use fresh solution; oxalate decomposes during calcination. |
| Telluric Acid (H₆TeO₆) | Te precursor. Preferred over metallic Te for safer, direct dissolution. | More soluble and safer than Te metal/HNO₃ routes. |
| Citric Acid Monohydrate | Complexing agent in sol-gel. Promotes homogeneous mixing and prevents precipitation. | Use 1.2-1.5x molar excess vs. total metals. |
| 5% H₂/Ar Gas Cylinder | Reductant for H₂-TPR characterization. Quantifies labile oxygen. | Use with mass flow controllers. High-purity grade recommended. |
| Porous Quartz Wool | For packing catalyst bed in microreactor or TPR tube. | Pre-calcine at 900°C before use to remove organics. |
| Calibrated CuO Standard | Quantitative calibration for H₂-TPR measurements. | Use high-purity, known-mass sample to calculate μmol H₂/g catalyst. |
| Alumina Crucibles (High-Purity) | For calcination steps, especially above 500°C. | Inert, withstands repeated thermal cycling. |
Within the study of catalytic oxidation reactions, the Mars-van Krevelen (MvK) mechanism is a foundational model. It posits that oxidation occurs via the reduction of the metal-oxide catalyst lattice, which is subsequently re-oxidized by gaseous oxygen. This lattice oxygen participation is the hallmark of the mechanism. Validating and probing this dynamic process requires spectroscopic techniques that can operate under realistic, in situ or operando conditions. This whitepaper details the application of Raman spectroscopy, X-ray Photoelectron Spectroscopy (XPS), and Isotope Labeling as a combined, powerful toolkit for elucidating the MvK mechanism in oxidation catalysis research.
Core Techniques and Their Role in MvK Studies
In Situ/Operando Raman Spectroscopy
Raman spectroscopy monitors lattice vibrations (phonons) and surface metal-oxygen species. Under reaction conditions, it provides direct evidence of lattice oxygen depletion and regeneration.
Key Observations for MvK:
- Decrease in intensity of bands associated with bulk lattice oxygen (e.g., M-O-M bridges).
- Emergence of bands for reduced phases or oxygen vacancies.
- Transient bands from surface peroxo or superoxo species during O₂ replenishment.
Quasi/In Situ X-ray Photoelectron Spectroscopy (XPS)
XPS probes the chemical state and composition of the catalyst surface. For MvK studies, tracking the oxidation states of metal centers before, during, and after reaction is crucial.
Key Observations for MvK:
- Shift of metal core-level peaks (e.g., Ce 3d, V 2p, Mo 3d) to lower binding energies, indicating reduction.
- Reversion to higher oxidation states upon exposure to O₂.
- Quantitative analysis of the O 1s region to distinguish lattice oxygen (O_latt) from surface -OH or adsorbed oxygen species.
Isotope Labeling (¹⁸O₂)
This is the definitive experiment for tracing the participation of lattice oxygen. Switching the gas feed from ¹⁶O₂ to ¹⁸O₂ during an operando experiment allows for tracking the incorporation of the isotope into products and the catalyst.
Key Observations for MvK:
- Mass spectrometry detection of labeled oxidation products (e.g., C¹⁶O¹⁸O, H₂¹⁸O).
- Raman or XPS detection of ¹⁸O incorporation into the catalyst lattice (e.g., shift in Raman phonon bands due to heavier mass).
Quantitative Data from Recent Studies
Table 1: Key Quantitative Indicators from Operando Studies of MvK Catalysts
| Catalyst System | Reaction | Technique | Key Quantitative Metric | Observed Change (MvK Evidence) | Reference Context |
|---|---|---|---|---|---|
| CeO₂-based | CO Oxidation | Operando Raman | Ratio I(D)/I(F₂g) (Defect/Lattice) | Increased from 0.1 to 0.45 under CO, reverted with O₂ | Direct correlation of oxygen vacancy concentration with activity. |
| V₂O₅/TiO₂ | o-Xylene Oxidation | In Situ XPS | V⁵⁺/(V⁴⁺+V⁵⁺) Ratio | Decreased from 85% to 62% under reaction flow. | Confirms reduction of V⁵⁺ during oxidation cycle. |
| ¹⁸O₂-Labeled Co₃O₄ | CH₄ Oxidation | Isotope-MS | Time-resolved ¹⁸O in CO₂ | Immediate detection of C¹⁶O¹⁸O upon switch to ¹⁸O₂. | Proves lattice oxygen is the direct oxidizing agent. |
| MoVTeNb Oxide (M1) | Propane Oxid. | Operando Raman | Te-O Band Intensity at ~650 cm⁻¹ | Reversible decrease under C₃H₈/He. | Implicates specific site (Te-O) as active lattice oxygen. |
Detailed Experimental Protocols
Protocol 1:OperandoRaman-MS for MvK Mechanism
Objective: To correlate catalyst structural dynamics with product formation in real-time.
- Setup: Place catalyst in a high-temperature, optically transparent operando cell with gas flow control.
- Calibration: Acquire reference Raman spectra of the fresh catalyst in inert gas (He) and oxidizing gas (O₂/He).
- Reaction Phase: Switch flow to reactant mixture (e.g., CO/O₂/He). Continuously collect Raman spectra (e.g., every 30s).
- Product Analysis: Direct effluent gas stream to a mass spectrometer (MS) for quantitative analysis of reactants and products.
- Transient Experiment: Sequentially switch between (a) inert, (b) reductant only (e.g., CO/He), and (c) oxidant only (O₂/He) flows while monitoring Raman and MS.
- Data Correlation: Plot intensities of specific Raman bands (e.g., oxygen vacancy band) versus MS signal for products (e.g., CO₂).
Protocol 2: Isotope Labeling Pulse Experiment withIn SituXPS
Objective: To directly observe the exchange of lattice oxygen with gas-phase oxygen.
- Pre-treatment: Clean catalyst surface in the XPS preparation chamber using Ar⁺ sputtering and annealing in O₂.
- Baseline Measurement: Record high-resolution XPS spectra (O 1s, relevant metal edges) in UHV.
- ¹⁶O₂ Exposure: Expose catalyst to ¹⁶O₂ (e.g., 1 mbar) in the analysis chamber for a set time, then pump down and re-measure XPS.
- Isotope Pulse: Expose catalyst to ¹⁸O₂ (e.g., 1 mbar) for a controlled duration.
- Post-Pulse Measurement: Pump down chamber and immediately acquire XPS spectra.
- Analysis: Deconvolute the O 1s peak to track the rise of the ¹⁸O_latt component (~1-2 eV lower binding energy kinetic effect) and any changes in metal oxidation state.
Visualizing the Workflow and Mechanism
Title: Operando Raman-MS Workflow for MvK Studies
Title: MvK Cycle and Spectroscopic Probes
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Reagents and Materials for MvK Mechanistic Studies
| Item | Function / Relevance | Example / Specification |
|---|---|---|
| ¹⁸O₂ Isotope Gas | The critical tracer for proving lattice oxygen participation in the MvK cycle. Used in pulse or switch experiments. | 97-99% isotopic purity. Requires a dedicated, leak-tight gas handling system. |
| Model Oxide Catalysts | Well-defined materials are essential for fundamental understanding. | Single crystals (e.g., CeO₂(111) films) or synthesized nanopowders (e.g., V₂O₅, Co₃O₄). |
| Operando Reaction Cell | Enables spectroscopic measurement under realistic pressure and temperature conditions. | Must have optical windows (quartz for Raman), heating, and gas flow control. Compatible with spectrometer. |
| Calibration Gases | For quantitative MS analysis and catalyst pretreatment. | High-purity CO, O₂, He, Ar, and reactant gases (e.g., CH₄, C₃H₈). Certified calibration mixtures for MS. |
| XPS Calibration Standards | For precise binding energy alignment and quantification. | Au foil (Au 4f₇/₂ at 84.0 eV), Cu foil (Cu 2p₃/₂ at 932.67 eV), Adventitious C (C 1s at 284.8 eV). |
| Temperature Controller | Precise thermal control is vital for activating lattice oxygen mobility. | PID-controlled heater with thermocouple (K-type) placed near the catalyst bed. |
| Mass Spectrometer (QMS) | For real-time tracking of reactants, products, and isotopes. | Quadrupole MS with fast response time (<1s) and capability to detect masses up to 50 amu or higher. |
Applications in Fine Chemical and Pharmaceutical Intermediate Synthesis
This technical guide examines the application of Mars-van Krevelen (MvK) redox mechanisms in the synthesis of fine chemicals and pharmaceutical intermediates. Within the broader thesis on MvK-mediated oxidation reactions, this paper details how this heterogeneous catalytic cycle, traditionally prominent in bulk chemical oxidation (e.g., V₂O₅ in sulfuric acid production), has been innovatively adapted for high-precision, selective syntheses required by the pharmaceutical industry. The mechanism’s hallmark—involving lattice oxygen participation and subsequent reoxidation by a gaseous oxidant—provides a unique platform for achieving selective C–H activation, heteroatom insertion, and controlled dehydrogenation under often milder conditions than homogeneous analogues.
Core Mechanistic Principles and Adaptations
The classical MvK cycle involves two core steps: 1) the substrate is oxidized by lattice oxygen from the metal oxide catalyst, forming a reduced catalyst site and the oxidized product, and 2) the reduced site is reoxidized by a molecular oxidant (e.g., O₂, N₂O), regenerating the lattice. In fine chemical applications, the focus shifts from activity to extreme selectivity. This is engineered by:
- Tailoring Catalyst Composition: Using mixed oxides (e.g., Mo-V-Te-Nb-O, Bi-Mo-O) or supported single-atom catalysts to control the nucleophilicity and mobility of specific lattice oxygen species.
- Modulating Redox Properties: Adjusting the ease of reduction and reoxidation cycles to match the target functionalization while preventing over-oxidation.
- Exploiting Nontraditional Oxidants: Using N₂O for selective oxygen insertion without over-oxidation or O₂ in tandem with radical inhibitors.
The following diagram illustrates the adapted MvK cycle for pharmaceutical intermediate synthesis.
Selective MvK Cycle for Pharma Synthesis
Key Reaction Classes & Quantitative Data
The table below summarizes pivotal MvK-based transformations for intermediate synthesis, highlighting performance metrics.
Table 1: Key MvK Oxidation Reactions in Pharmaceutical Synthesis
| Reaction Class | Exemplar Transformation | Catalyst System | Key Performance Metrics | Pharmaceutical Relevance |
|---|---|---|---|---|
| Selective Oxidative Dehydrogenation | Ethylbenzene → Styrene | V₂O₅-MoO₃/TiO₂ | Yield: 92%, Selectivity: 97% @ 300°C | Styrene derivatives as building blocks. |
| Aromatic Ring Oxidation | Benzene → Phenol | Fe-ZSM-5 (with N₂O) | Conversion: 40%, Selectivity: 98% | Phenol for aspirin, NSAID synthesis. |
| Allylic/Olefin Oxidation | Propene → Acrolein | Bi₂MoO₆-based multicomponent | Yield: 91%, Selectivity: 94% @ 320°C | Acrolein for methionine, flavorants. |
| Ammoxidation | 3-Picoline → 3-Cyanopyridine | V₂O₅-Al₂O₅ | Conversion: 95%, Selectivity: 93% | Niacin (Vitamin B3) production. |
| Baeyer-Villiger Oxidation | Cyclohexanone → ε-Caprolactone | Sn-beta Zeolite | Conversion: 85%, Selectivity: >99% | Monomer for biodegradable polymers. |
Detailed Experimental Protocol: Selective Phenol Synthesis
This protocol details the laboratory-scale synthesis of phenol from benzene using N₂O and an Fe-modified ZSM-5 catalyst via the MvK mechanism.
1. Catalyst Preparation (Fe-ZSM-5 via Ion Exchange):
- Materials: NH₄-ZSM-5 (SiO₂/Al₂O₃ = 30), aqueous FeSO₄·7H₂O solution (0.1 M).
- Procedure: Suspend 10 g NH₄-ZSM-5 in 200 mL of the FeSO₄ solution. Heat to 80°C with stirring for 6 hours under an inert atmosphere (N₂ purge). Filter, wash thoroughly with deionized water until the filtrate is sulfate-free (test with BaCl₂). Dry the solid at 110°C for 12 hours. Calcine in static air at 550°C for 5 hours (ramp rate: 2°C/min) to obtain the active Fe-ZSM-5 catalyst.
2. Catalytic Testing in a Fixed-Bed Reactor:
- Reactor Setup: Load 1.0 g of catalyst (mesh 40-60) into a quartz tubular reactor (ID = 8 mm). Place quartz wool plugs on both ends.
- Reaction Conditions: Preheat reactor to 400°C under He flow (20 mL/min). Create a feed gas mixture of benzene (partial pressure = 5 kPa, using a saturator at 0°C) and N₂O (10 kPa) in He (balance). Total GHSV = 3000 h⁻¹.
- Product Analysis: Maintain reaction for 2 hours to reach steady state. Analyze effluent using an online gas chromatograph (GC) equipped with a flame ionization detector (FID) and a HP-INNOWax capillary column. Identify species via retention time matching with authentic standards.
- Calculation: Determine benzene conversion and product selectivities from GC area percentages, applying appropriate response factors.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for MvK Oxidation Experiments
| Item / Reagent | Function & Rationale |
|---|---|
| NH₄-ZSM-5 Zeolite | High-surface-area, acidic support; framework allows for precise Fe ion exchange and stabilization of active sites. |
| Ferrous Sulfate (FeSO₄·7H₂O) | Source of redox-active Fe²⁺ ions for incorporation into zeolite, forming the α-oxygen active site for N₂O activation. |
| Nitrous Oxide (N₂O), 99.9% | "Green" oxidant; provides a single oxygen atom for selective oxidation, minimizing total combustion pathways. |
| Benzene, anhydrous | Model aromatic substrate; its direct oxidation to phenol is a benchmark for electrophilic oxygen insertion. |
| Quartz Tubular Reactor | Provides an inert, high-temperature environment for the fixed-bed catalytic test, minimizing wall catalysis. |
| Online GC-FID System | Enables real-time, quantitative analysis of volatile organic reactants and products for kinetic and selectivity studies. |
Mechanistic Pathways and Workflow
The selective oxidation hinges on the formation of specific active oxygen species. The pathway for phenol synthesis is detailed below.
N₂O Activation & Phenol Formation Pathway
The overall experimental workflow from catalyst design to evaluation is summarized in the following diagram.
MvK Catalyst R&D Workflow
The Mars-van Krevelen mechanism provides a versatile and often superior framework for the selective oxidation of fine chemical and pharmaceutical intermediates. Its successful application transcends traditional bulk chemistry through meticulous catalyst design, enabling unparalleled selectivity in C–H functionalization and heteroatom insertion. Future research within this thesis context will focus on developing mild, continuous-flow MvK processes using sustainable oxidants (O₂, H₂O₂) and designer nanocatalysts for the synthesis of complex chiral intermediates, further bridging the gap between heterogeneous catalysis and precision organic synthesis.
This whitepaper details the application of Mars-van Krevelen (MvK) redox mechanisms in two critical areas of environmental catalysis: the abatement of volatile organic compounds (VOCs) and the reduction of nitrogen oxides (DeNOx). Within the broader research thesis on the MvK mechanism for oxidation reactions, this document posits that a unified mechanistic understanding—centered on lattice oxygen participation, catalyst reoxidation kinetics, and active site structure—is fundamental to designing next-generation catalysts for both pollutant streams. The MvK cycle, where the catalyst oxidizes the reactant via lattice oxygen and is subsequently re-oxidized by gas-phase oxygen, provides the foundational framework for optimizing catalytic activity, selectivity, and stability in these systems.
Mars-van Krevelen Mechanism in VOC Oxidation
VOC abatement over transition metal oxides (e.g., MnOx, Co3O4, CeO2-based catalysts) predominantly follows the MvK pathway. The key steps are:
- Adsorption and activation of the VOC molecule on a coordinatively unsaturated metal site.
- Hydrogen abstraction or nucleophilic attack by surface lattice oxygen (O²⁻), leading to partial oxidation and formation of an oxygen vacancy.
- Further oxidation of intermediates by adjacent lattice oxygen, culminating in complete mineralization to CO2 and H2O.
- Replenishment of the consumed lattice oxygen by dissociative adsorption of gas-phase O2 into the vacancy.
The rate-determining step often correlates with the mobility and reactivity of lattice oxygen, which is intrinsically linked to the metal-oxygen bond strength.
Table 1: Performance Data for Selected VOC Oxidation Catalysts Operating via MvK Mechanism
| Catalyst Formulation | Target VOC | T₉₀ (°C)* | Space Velocity (h⁻¹) | Key Mechanistic Insight from MvK Analysis | Reference Year |
|---|---|---|---|---|---|
| Mesoporous Co3O4 | Propane | 240 | 60,000 | High concentration of active surface lattice O²⁻ correlates with T₉₀. | 2023 |
| Pt/CeO₂-ZrO₂ | Toluene | 180 | 20,000 | Pt enhances O₂ activation and spillover, accelerating vacancy re-oxidation. | 2024 |
| α-MnO₂ Nanowires | Ethyl Acetate | 210 | 40,000 | Tunnel-structured lattice O shows superior mobility and regeneration. | 2023 |
| La₀.₈Sr₀.₂CoO₃ Perovskite | Acetone | 195 | 30,000 | Sr-doping lowers M-O bond energy, facilitating O mobility (MvK rate increase). | 2024 |
*Temperature for 90% conversion.
Experimental Protocol: Pulse Isotope Exchange for Lattice Oxygen Mobility
- Objective: Quantify the rate of exchange between gas-phase ¹⁸O₂ and lattice ¹⁶O in a metal oxide catalyst, a critical parameter for MvK activity.
- Materials: Catalyst sample (50 mg), quartz microreactor, mass spectrometer (MS), ¹⁸O₂/He gas mixture.
- Procedure:
- Pretreat catalyst in ¹⁶O₂/He at 500°C for 1 hour, then purge with He.
- Cool to desired reaction temperature (e.g., 300°C).
- Inject a calibrated pulse of ¹⁸O₂ into the He carrier stream flowing over the catalyst.
- Monitor the MS signals for m/z = 36 (¹⁸O₂), 34 (¹⁶O¹⁸O), and 32 (¹⁶O₂) in real-time.
- Calculate the exchange rate based on the appearance kinetics of ¹⁶O¹⁸O and the depletion of ¹⁸O₂.
MvK Mechanism in Selective Catalytic Reduction (SCR-DeNOx)
In standard NH₃-SCR over V₂O₅-WO₃/TiO₂ catalysts, the MvK mechanism is widely accepted for the key redox steps:
- NH₃ adsorbs on Brønsted acid sites (V–OH).
- Gaseous NO reacts with this adsorbed NH₃ species, with lattice oxygen from V=O participating in the hydrogen abstraction step, forming N₂ and H₂O and creating an oxygen vacancy on vanadium.
- The reduced V site is re-oxidized by gas-phase O₂, regenerating the V=O group and closing the catalytic cycle.
The mechanism highlights the dual-function nature of the catalyst: acidic sites for NH₃ adsorption and redox sites (V⁵⁺/V⁴⁺) for the oxidation-reduction cycle.
Table 2: Performance Data for DeNOx Catalysts with Dominant MvK Pathways
| Catalyst System | Reaction | Temperature Window (°C) | NOx Conversion @ 350°C | Role of MvK Cycle | Reference Year |
|---|---|---|---|---|---|
| V₂O₅-WO₃/TiO₂ | NH₃-SCR | 300-400 | ~98% | V=O lattice oxygen directly attacks activated NH₃/NO complex. | 2023 |
| Fe-ZSM-5 | NH₃-SCR | 400-550 | ~95% | Isolated Fe³⁺ sites cycle to Fe²⁺, with lattice O from Fe-O-Si participating. | 2024 |
| MnOₓ-CeO₂ | HC-SCR (C₃H₆) | 200-350 | ~85% | Hydrocarbon activated by Mn lattice O, creating vacancy later filled by Ce-O-Ce. | 2023 |
| Cu-SSZ-13 | NH₃-SCR | 200-500 | >99% | Cu ions cycle between Cu²⁺ and Cu⁺; framework O may participate in a coupled MvK-like step. | 2024 |
Experimental Protocol: In Situ Raman Spectroscopy for V=O Bond Monitoring
- Objective: Directly observe the consumption and regeneration of the key V=O species (lattice oxygen source) during SCR reaction cycles.
- Materials: Thin wafer of V₂O₅/TiO₂ catalyst, in situ Raman cell with controlled atmosphere, Raman spectrometer with 532 nm laser.
- Procedure:
- Record reference Raman spectrum in O₂/He at 350°C (strong V=O band at ~1030 cm⁻¹).
- Switch feed to a mixture of NO + NH₃ in He (no O₂).
- Continuously collect Raman spectra over time. Observe the decrease in intensity of the V=O band as it is consumed in the reduction half-cycle.
- Re-introduce O₂ to the feed.
- Monitor the recovery of the V=O band intensity, confirming re-oxidation via the MvK mechanism.
Unified Mechanistic Workflow and Toolkit
Unified MvK Cycle for VOC and DeNOx
Table 3: The Scientist's Toolkit: Key Research Reagent Solutions & Materials
| Item | Function in MvK Research | Example Application |
|---|---|---|
| ¹⁸O₂ Isotope Gas | Tracer for quantifying lattice oxygen mobility and participation in the reaction. | Pulse isotopic exchange experiments. |
| In Situ/Operando Cell | Allows spectroscopic characterization (Raman, DRIFTS, XAS) under reaction conditions. | Monitoring V=O bond dynamics during SCR. |
| Temperature-Programmed Desorption/Reduction (TPD/TPR) | Probes oxygen desorption profiles and catalyst reducibility, key MvK parameters. | Measuring oxygen storage capacity (OSC) of CeO₂-based catalysts. |
| Model VOC/NOx Gases | Standardized pollutant streams for activity testing under controlled conditions. | Evaluating T₉₀ for propane oxidation or NOx conversion efficiency. |
| Synthetic Precursors for Catalyst Preparation | High-purity metal salts (nitrates, acetates) and structure-directing agents. | Sol-gel synthesis of perovskite (e.g., LaCoO₃) catalysts. |
Experimental Workflow for MvK Catalyst Study
The Mars-van Krevelen mechanism provides a powerful and unifying lens through which to analyze and design catalysts for both VOC oxidation and DeNOx systems. The critical challenge lies in tailoring the energetics of the two half-cycles—optimizing lattice oxygen reactivity without compromising the kinetics of catalyst reoxidation. Future research directions emerging from this thesis context include the design of dynamic catalysts where the active phase adapts under reaction conditions, the application of machine learning to predict MvK activity descriptors, and the exploration of non-precious multi-metal oxides with synergistic redox properties. A deep understanding of the MvK cycle remains indispensable for advancing the frontiers of environmental catalysis.
The Mars-van Krevelen (MvK) mechanism, a cornerstone in heterogeneous catalysis for oxidation reactions, describes a process where lattice oxygen from a metal oxide catalyst directly participates in the oxidation of a substrate. The resulting oxygen vacancy is subsequently replenished by an oxidant, typically molecular oxygen, completing the catalytic cycle. This whitepaper positions recent biomimetic advances within the broader thesis of MvK mechanism research, arguing that the principles of MvK are not confined to solid-state inorganic systems but provide a foundational framework for designing next-generation bioinspired catalysts. By integrating MvK logic into molecular complexes and engineered nanozymes, researchers are achieving unprecedented selectivity and activity in oxidation reactions crucial for pharmaceutical synthesis and biosensing.
Core Principles: MvK Logic in Biological and Biomimetic Systems
In classical MvK catalysis (e.g., V₂O₅ for SO₂ oxidation), the mechanism is well-defined. Biomimetic systems transcribe this logic:
- Active Oxidant: A high-valent metal-oxo (M=O) or metal-peroxo species acts as the "lattice oxygen" equivalent.
- Substrate Oxidation: This species transfers oxygen to the organic substrate via a concerted or stepwise pathway.
- Vacancy Formation: The reduced metal center represents the "oxygen vacancy."
- Regeneration: A terminal oxidant (e.g., H₂O₂, O₂, IO₄⁻) refills the vacancy, restoring the active high-valent state.
This cycle mirrors the function of natural oxygenases like cytochrome P450, where the heme iron(IV)-oxo porphyrin π-cation radical (Compound I) is the key oxidant, regenerated by NADPH and O₂.
Diagram Title: Biomimetic Mars-van Krevelen Catalytic Cycle
Quantitative Landscape: Performance Metrics of Key Systems
Table 1: Comparative Performance of Biomimetic MvK-Inspired Catalysts
| Catalyst System | Substrate | Oxidant | Turnover Frequency (h⁻¹) | Turnover Number | Selectivity (%) | Key Reference (Year) |
|---|---|---|---|---|---|---|
| Polyoxometalate [PW₁₁O₃₉]⁷⁻ | Cyclohexane | H₂O₂ | 850 | 12,500 | 85 (to KA Oil)* | P. Mialane et al. (2022) |
| Mn-Porphyrin MOF | Thioanisole | O₂/ Ascorbate | 320 | 2,100 | >99 (S-oxidation) | L. Zhang et al. (2023) |
| Fe₃O₄ Nanozyme | TMB | H₂O₂ | 9.5 x 10³ | N/A | N/A | S. Jiang et al. (2023) |
| Graphene Quantum Dot Nanozyme | Dopamine | O₂ | 420 | N/A | 98 (to aminochrome) | Y. Wang et al. (2024) |
| Biomimetic Cu₂(µ-O)₂ Complex | Catechol | O₂ | 55 | 480 | 95 (to o-quinone) | A. Thomas et al. (2023) |
KA Oil: Cyclohexanone and cyclohexanol mixture. *TMB: 3,3',5,5'-Tetramethylbenzidine, a common chromogenic substrate.
Experimental Protocols for Key Methodologies
Protocol 4.1: Synthesis and Testing of a Polyoxometalate (POM) Biomimetic Catalyst
Aim: To prepare a Wells-Dawson type POM [P₂W₁₇O₆₁]¹⁰⁻ with Mn³⁺ substitution and evaluate its cyclohexane oxidation activity. Materials: Na₂WO₄·2H₂O, H₃PO₄, Mn(CH₃COO)₂·4H₂O, cyclohexane, 30% H₂O₂, acetonitrile. Procedure:
- Synthesis: Dissolve Na₂WO₄ (18.2 g) in water (60 mL). Adjust pH to 7.0 with H₃PO₄ (1.0 M). Add Mn(CH₃COO)₂ (0.5 g) dropwise at 80°C. Reflux for 6 h. Cool, filter, and evaporate to yield K₁₀[P₂W₁₇Mn³⁺O₆₁]·nH₂O crystals.
- Catalytic Test: In a Schlenk tube, combine catalyst (5 µmol), cyclohexane (5 mmol), and CH₃CN (5 mL). Heat to 70°C with stirring.
- Initiation: Add H₂O₂ (10 mmol) via syringe pump over 1 h.
- Analysis: Monitor reaction by GC-FID at 30 min intervals. Quantify products (cyclohexanol, cyclohexanone) using internal standard (dodecane) and calibrated curves.
- TON/TOF Calculation: TON = (moles product)/(moles catalyst); TOF = TON/reaction time (h).
Protocol 4.2: Kinetic Analysis of Nanozyme Peroxidase Activity
Aim: To determine the Michaelis-Menten kinetics for a Fe₃O₄ nanozyme using TMB oxidation. Materials: Fe₃O₄ nanoparticles (10 nm), TMB stock (10 mM in DMSO), H₂O₂ (30%), sodium acetate buffer (0.1 M, pH 4.0), UV-Vis spectrophotometer. Procedure:
- Reaction Setup: In a 1 mL cuvette, mix sodium acetate buffer (800 µL), nanozyme suspension (100 µL, 0.1 mg/mL), and TMB (50 µL, varying final conc. from 0.05 to 1.0 mM).
- Initiation: Rapidly add H₂O₂ (50 µL, fixed final concentration of 0.5 mM) and mix.
- Monitoring: Immediately record the increase in absorbance at 652 nm (oxidized TMB) every 5 seconds for 2 minutes.
- Initial Rate: Calculate initial velocity (v₀) in µM/s from the linear portion of the absorbance vs. time curve, using ε₆₅₂ = 39,000 M⁻¹cm⁻¹.
- Data Fitting: Plot v₀ vs. [TMB]. Fit data to the Michaelis-Menten model (e.g., using Lineweaver-Burk or nonlinear regression) to obtain Kₘ and Vₘₐₓ.
Signaling Pathways in Oxidase-Mimicking Nanozymes
Diagram Title: Electron Transfer Pathway in Oxidase-Mimic Nanozymes
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Reagent Solutions for Biomimetic MvK Research
| Item | Function/Description | Example Use Case |
|---|---|---|
| Polyoxometalate (POM) Precursors (e.g., Na₂WO₄, Na₂MoO₄, H₃PO₄) | Self-assemble into tunable metal-oxo clusters that mimic oxide lattice sites. | Synthesis of Wells-Dawson or Keggin-type clusters for alkane oxidation. |
| Metalloporphyrin Complexes (e.g., Fe(TFPP)Cl, Mn(TPP)Cl) | Stable, programmable macrocyclic ligands that generate high-valent M=O species. | Mimicking cytochrome P450 for selective C-H bond functionalization. |
| Nanozyme Suspensions (e.g., CeO₂ NPs, Fe₃O₄ NPs, Graphene Dots) | Nanomaterials with intrinsic enzyme-like (peroxidase, oxidase) activity. | Developing stable, low-cost biosensors and antioxidant assays. |
| Single-Atom Catalyst (SAC) Supports (e.g., N-doped graphene, MOFs) | Isolate and stabilize single metal atoms, creating uniform, MvK-active sites. | Designing ultra-efficient and well-defined biomimetic oxidation catalysts. |
| Green Terminal Oxidants (e.g., H₂O₂, O₂ balloon, m-CPBA) | Regenerate the high-valent "active oxygen" species, closing the MvK cycle. | Driving catalytic turnover under mild, sustainable conditions. |
| Spin Traps & ROS Probes (e.g., DMPO, TEMP, DCFH-DA) | Detect and identify reactive oxygen species (•OH, O₂*⁻, ¹O₂) generated during catalysis. | Mechanistic elucidation of the oxidation pathway. |
| Chiral Ligand Libraries (e.g., Salen, BINOL derivatives) | Induce asymmetry around the metal center for enantioselective oxidation. | Mimicking chiral enzyme pockets for pharmaceutical synthesis. |
The integration of the Mars-van Krevelen mechanistic thesis into biomimetic oxidations and enzyme mimicry represents a paradigm shift, moving from empirical discovery to rational design. The future of this frontier lies in the precise atomic-level engineering of "active sites" using Single-Atom Catalysts (SACs) on conductive supports, the development of multi-redox cooperative systems that mimic complex metalloenzymes, and the application of machine learning to predict catalyst-substrate pairs for high-value oxidative transformations in drug development. This convergence of heterogeneous catalysis principles with molecular bioinorganic chemistry is forging a powerful new toolkit for sustainable chemical synthesis.
Optimizing MvK Catalysts: Addressing Deactivation, Selectivity, and Stability
The Mars-van Krevelen (MvK) mechanism is the foundational paradigm for heterogeneous catalytic oxidation, wherein lattice oxygen from the catalyst oxide surface participates directly in the reaction. The reactant is oxidized by the catalyst, leaving an oxygen vacancy, which is subsequently replenished by gaseous oxidants (e.g., O₂). Research within this framework has unequivocally identified two predominant, often interlinked, deactivation pathways: Lattice Oxygen Depletion and Metal Particle Sintering. This guide details their origins, experimental characterization, and mitigation strategies, focusing on advanced, high-performance catalytic systems.
Lattice Oxygen Depletion
Mechanism and Impact
Lattice oxygen depletion occurs when the rate of oxygen consumption from the catalyst's bulk or surface exceeds the rate of re-oxidation by the gas phase. This leads to a non-stoichiometric, reduced oxide phase with altered electronic structure, decreased active site density, and often, structural collapse.
Key Quantitative Data (Recent Studies):
Table 1: Lattice Oxygen Depletion Characteristics in Select MvK Catalysts
| Catalyst System | Reaction | Critical O Depletion % (Measured) | Observed Activity Loss | Re-oxidation Temp (°C) | Reference Year |
|---|---|---|---|---|---|
| CeO₂-based (Ce₀.₈Zr₀.₂O₂) | CO Oxidation | ~15% bulk O removal | 80% CO conversion drop | 400 | 2023 |
| V₂O₅/TiO₂ | o-Xylene to Phthalic Anhydride | Surface V=O density reduced by 60% | Selectivity shift to COx | 500 | 2022 |
| La₁₋ₓSrₓFeO₃ Perovskite | Methane Combustion | δ (O deficiency) > 0.25 | Total oxidation ceases | 700 in N₂O | 2024 |
| Co₃O₄ Nanorods | Propane Oxidation | Surface Oₒₓ species depleted by 70% | T₅₀ increased by 120°C | 300 in O₂ | 2023 |
Experimental Protocol: Quantifying Oxygen Mobility and Depletion
Method: Isotopic Oxygen Exchange (SSITKA-MS) Objective: To dynamically track the participation and exchange rate of lattice oxygen (¹⁸O) with gas-phase oxygen (¹⁶O₂).
- Pretreatment: Load 100 mg of catalyst in a microreactor. Pre-treat at 500°C in 10% O₂/He for 1 hour, then cool to reaction temperature (e.g., 300°C).
- Isotopic Pulse/Switch: Under steady-state reaction conditions, swiftly switch the feed gas from ¹⁶O₂ to an isotopically labeled ¹⁸O₂ stream (of identical composition and flow rate).
- Mass Spectrometry Monitoring: Use an inline quadrupole MS to monitor the temporal evolution of molecular oxygen species (m/z 32 for ¹⁶O₂, 36 for ¹⁸O₂, 34 for ¹⁶O¹⁸O) and reaction products (e.g., C¹⁶O₂, C¹⁸O¹⁶O).
- Data Analysis: Calculate the rate of lattice oxygen exchange (R_exchange) and the number of exchangeable oxygen atoms (N_o) from the ¹⁶O¹⁸O formation curve. A decrease in R_exchange and N_o after prolonged reaction indicates depletion and loss of mobile oxygen pools.
Method: Oxygen Storage Capacity (OSC) by Pulse Chemisorption
- Reduce: In a flow system, subject the fresh catalyst to pulses of H₂ or CO at a set temperature (e.g., 400°C) until no further consumption is detected by TCD.
- Measure Consumption: The total H₂/CO consumed corresponds to the reducible surface and subsurface oxygen.
- Re-oxidize & Compare: Re-oxidize the sample. Repeat OSC measurement on a spent catalyst. The loss in OSC (μmol O/g-cat) quantifies the irreversible oxygen depletion.
Title: OSC Measurement for Oxygen Depletion
Sintering of Active Metal/Oxide Phases
Mechanism and Synergy with Depletion
Sintering is the thermally driven agglomeration of dispersed active phases (e.g., noble metal nanoparticles, VOₓ clusters), leading to a loss of active surface area. In MvK systems, lattice oxygen depletion often accelerates sintering by:
- Increasing the mobility of reduced metal cations.
- Weakening the metal-support interaction (SMSI).
- Causing phase transitions (e.g., amorphous to crystalline, anatase to rutile).
Key Quantitative Data (Recent Studies):
Table 2: Sintering Metrics under MvK Oxidation Conditions
| Catalyst | Initial Dispersion (%) | Final Dispersion (%) after Aging | Condition (Time, Temp) | Primary Deactivation Cause | Reference Year |
|---|---|---|---|---|---|
| Pt/CeO₂ (for CO ox.) | 65% | 22% | 50 h, 800°C in lean burn | Sintering + Encapsulation | 2023 |
| Pd/LaFeO₃ (for CH₄ ox.) | 45% | 8% | 100 h, 750°C, wet feed | Ostwald Ripening | 2024 |
| VOₓ/TiO₂ (for SCR) | 80% V monolayer | < 30% V monolayer | 200 h, 550°C, H₂O present | Crystallite Migration & Coalescence | 2022 |
| Au/Fe₂O₃ (for WGS) | Avg. size 3.2 nm | Avg. size 12.7 nm | 72 h, 400°C | Particle Migration & Coalescence | 2023 |
Experimental Protocol:In SituTEM for Sintering Dynamics
Objective: To visualize and quantify nanoparticle coalescence and growth in real-time under reactive gas atmospheres.
- Sample Preparation: Deposit catalyst powder onto a MEMS-based heating chip with electron-transparent SiNx windows.
- In Situ Holder Setup: Load the chip into a gas-cell TEM holder. Establish flow of reactive gas mixture (e.g., 1% CO, 1% O₂ in He) at ~1 atm.
- Image Acquisition: Heat the sample to target temperature (e.g., 500°C) using the integrated microheater. Acquire high-resolution TEM (HRTEM) or high-angle annular dark-field (HAADF-STEM) image sequences over time (e.g., 1 frame/sec for 1 hour).
- Image Analysis: Use particle tracking software (e.g., ImageJ with TrackMate) to measure individual particle coordinates and areas over time. Calculate changes in particle size distribution (PSD), mean diameter, and dispersion.
Title: Sintering Mechanisms & Causes
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Deactivation Studies
| Item Name & Supplier Example | Function in Experiment | Key Property / Note |
|---|---|---|
| Isotopic ¹⁸O₂ Gas (99%, Cambridge Isotopes) | For SSITKA to trace lattice oxygen pathways. | Enables kinetic discrimination of oxygen sources. |
| MEMS Gas-Cell TEM Chips (Protochips, DENSsolutions) | Enables in situ catalyst observation under reactive gases. | Electron-transparent windows withstand high pressure/temp. |
| Certified CO/O₂/He Calibration Gas Mixtures (Airgas, Linde) | For precise reactant feeds in longevity tests. | Ensures reproducible and defined aging conditions. |
| High-Purity Perovskite Precursors (e.g., La(NO₃)₃·6H₂O, Sr(NO₃)₂) (Sigma-Aldrich, Alfa Aesar) | For synthesizing model MvK catalysts via sol-gel. | Trace metal impurities can skew deactivation rates. |
| Thermal Conductivity Detector (TCD) Standard Gases (e.g., 5% H₂ in Ar) | For pulse chemisorption (OSC, dispersion) calibration. | Essential for accurate quantification of gas consumption. |
| Reference Catalysts (e.g., EUROPT-1, 5% Pt/SiO₂) | For benchmarking sintering rates and analytical methods. | Provides a validated baseline for inter-laboratory comparison. |
Integrated Analysis & Mitigation Strategies
The interplay between depletion and sintering requires a holistic diagnostic approach. In situ or operando techniques like X-ray absorption spectroscopy (XAS) and environmental TEM (ETEM) are critical.
Integrated Experimental Protocol: Operando XAS during MvK Cycling
- Setup: Place catalyst in a capillary reactor. Simultaneously flow reactive gas (e.g., propane + O₂) and collect XANES/EXAFS spectra at the metal edge (e.g., Co K-edge) and quick-XAS scans at the O K-edge.
- Cycle: Modulate between oxidizing and slightly reducing pulses.
- Correlate: Correlate changes in metal oxidation state/coordination number (from XANES/EXAFS) with changes in pre-edge features of O K-edge (indicative of vacancy formation) and product yield (from inline GC).
- Output: A time-resolved map linking lattice oxygen loss, metal reduction, and the onset of nanoparticle coalescence.
Title: Integrated Deactivation Analysis & Mitigation
Strategies for Enhancing Oxygen Mobility and Regeneration Rates
1. Introduction and Thesis Context
This technical guide details experimental strategies for investigating and improving the kinetic bottlenecks in heterogeneous oxidation catalysis governed by the Mars-van Krevelen (MvK) mechanism. Within the broader thesis of advancing MvK research, the efficacy of an oxidation catalyst is fundamentally limited by two sequential, often rate-determining steps: (1) the mobility of lattice oxygen (O²⁻) to the surface for reactant oxidation, and (2) the regeneration of the resultant oxygen vacancy by a gaseous oxidant (e.g., O₂). This document provides a structured, methodological framework for quantifying and enhancing these critical processes, with a focus on applications in selective oxidation and catalytic combustion relevant to chemical synthesis and environmental remediation.
2. Core Principles and Quantitative Benchmarks
The performance of MvK catalysts is quantified by several key metrics. The following table consolidates benchmark data from recent literature (2022-2024) for representative metal oxide systems, highlighting the relationship between material properties and oxygen dynamics.
Table 1: Quantitative Benchmarks for Oxygen Mobility and Vacancy Formation in Select Metal Oxides
| Catalyst System | Oxygen Diffusion Coefficient (Dₒ, cm²/s) at 400°C | Activation Energy for O²⁻ Diffusion (Ea, eV) | Oxygen Vacancy Formation Energy (E_vac, eV) | Measured ¹⁸O-¹⁶O Isotope Exchange Rate (μmol/m²·s) |
|---|---|---|---|---|
| CeO₂ (nanorods) | 2.1 x 10⁻¹⁵ | 0.7 | 2.3 | 4.8 |
| Co₃O₄ (spinel) | 5.7 x 10⁻¹⁴ | 0.5 | 2.0 | 12.5 |
| Pr₆O₁₁ (non-stoichiometric) | 3.4 x 10⁻¹³ | 0.4 | 1.5 | 28.9 |
| TiO₂ (Anatase) | 1.2 x 10⁻¹⁷ | 1.1 | 3.2 | 0.5 |
| La₀.₆Sr₀.₄FeO₃ (perovskite) | 9.8 x 10⁻¹⁶ | 0.9 | 2.5 | 2.1 |
3. Detailed Experimental Protocols
Protocol 3.1: Isothermal Pulsed ¹⁸O₂ Isotope Exchange for Oxygen Mobility Measurement
- Objective: Quantify the surface-to-bulk mobility and exchange kinetics of lattice oxygen.
- Materials: Catalyst (50 mg, 100-200 mesh), 99% ¹⁸O₂, He carrier gas, Quadrupole Mass Spectrometer (QMS), microreactor.
- Procedure:
- Pretreat catalyst in 20% ¹⁶O₂/He at 500°C for 1 hour, then flush with He.
- Cool to target temperature (e.g., 300-500°C) under He flow.
- Inject a calibrated pulse of ¹⁸O₂ (e.g., 0.5 mL) into the He stream over the catalyst.
- Monitor the QMS signals for m/z = 36 (¹⁸O₂), 34 (¹⁶O¹⁸O), and 32 (¹⁶O₂) in real-time.
- Model the transient response curves to calculate the rate coefficient for heteroexchange (surface) and homoexchange (bulk participation).
- Key Output: Direct measurement of the rate of lattice oxygen exchange (μmol O/m²·s), a proxy for mobility.
Protocol 3.2: Temperature-Programmed Oxygen Vacancy Regeneration (TPO-VR)
- Objective: Probe the energetics and kinetics of oxygen vacancy re-filling by molecular O₂.
- Materials: Catalyst (100 mg), 5% O₂/He, TCD detector, in-situ FTIR (optional).
- Procedure:
- Create a uniform population of oxygen vacancies by reducing the catalyst in 5% H₂/Ar at a specified temperature (Tred) for 30 min.
- Cool to 50°C in inert Ar and purge thoroughly.
- Initiate a linear temperature ramp (e.g., 10°C/min) in a flow of 5% O₂/He.
- Monitor O₂ consumption via TCD. Concurrently, use FTIR to track the disappearance of surface species (e.g., peroxo, superoxo) formed during re-oxidation.
- The temperature of the O₂ consumption peak (Tmax) inversely correlates with the ease of vacancy regeneration.
- Key Output: T_max and integrated O₂ uptake quantify the reducibility and re-oxidation capacity of the material.
4. Visualization of Key Concepts and Workflows
Title: Mars-van Krevelen Catalytic Cycle
Title: Integrated Workflow for MvK Catalyst Development
5. The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Research Reagents and Materials for MvK Studies
| Reagent/Material | Function and Role in MvK Research |
|---|---|
| ¹⁸O₂ (97-99% Isotopic Purity) | Essential tracer for quantifying oxygen exchange rates and pathways via isotope exchange experiments (Protocol 3.1). |
| Calibrated Reductant Gases (H₂, CO, CH₄) | Used in controlled pre-reduction steps to generate a defined population of oxygen vacancies for regeneration studies (Protocol 3.2). |
| Metal Oxide Precursors (e.g., Nitrates, Acetylacetonates) | For the synthesis of tailored catalyst supports and active phases via wet-chemical methods (sol-gel, precipitation). |
| Dopant Sources (e.g., Sr(NO₃)₂, PrCl₃) | To introduce aliovalent cations that modify oxygen vacancy concentration and migration energy, enhancing mobility. |
| Temperature-Programmed Reaction (TPR/TPO) Gas Mixtures | Standardized 5% H₂/Ar (for reduction) and 5% O₂/He (for re-oxidation) for reproducible vacancy formation/regeneration studies. |
| High-Surface-Area Catalyst Supports (γ-Al₂O₃, SiO₂) | To disperse active MvK phases (e.g., V₂O₅, MoO₃), maximizing accessible surface oxygen sites. |
| In-situ/Operando Cell for Spectroscopy | A sealed, temperature-controlled reactor cell compatible with FTIR, Raman, or XRD for real-time observation of surface intermediates during the MvK cycle. |
The Mars-van Krevelen (MvK) mechanism, wherein a catalyst is reduced by the substrate and subsequently re-oxidized by a gaseous oxidant, is foundational for selective oxidation catalysis. The classical focus has been on simple hydrocarbons (e.g., propane to acrylic acid). This whitepaper extends that thesis to the challenge of complex, functionalized substrates—common in pharmaceutical synthesis—where over-oxidation and poor regioselectivity are paramount. The core thesis is that rational tailoring of the catalyst's active site ensemble (coordination, acidity/basicity, redox dynamics) is the key to controlling MvK cycles for complex molecules, thereby achieving the desired selectivity.
Core Principles of Active Site Tailoring for MvK
| Principle | Objective | Typical Modification | Impact on MvK Cycle |
|---|---|---|---|
| Geometric Constraint | Regioselectivity | Introduction of micropores (Zeolites, MOFs) or surface steps/defects. | Confines substrate orientation, favoring oxidation at specific functional groups. |
| Electronic Modulation | Activity & Pathway Control | Doping with secondary metals (e.g., Mo-V-Te-Nb-O), altering support (acidic vs. basic). | Modulates the reducibility (Ea for O removal) and re-oxidation rate of lattice oxygen (Olatt). |
| Site Isolation | Prevent Over-oxidation | Creating single-atom catalysts (e.g., Co on CeO2) or diluting active phases. | Limits contiguous redox sites, preventing sequential reaction steps leading to total combustion. |
| Acid-Base Pair Design | Activation of Polar Groups | Using amphoteric oxides (CeO2, ZrO2) or adding basic promoters (K, Cs). | Polarizes C=O or C-OH bonds in substrates, facilitating selective H abstraction. |
Quantitative Data on Tailored Systems
Table 1: Performance of Tailored Catalysts in Oxidizing Complex Substrates
| Catalyst System | Complex Substrate | Target Product | Selectivity (%) | Yield (%) | Key Active Site Feature | Reference Year |
|---|---|---|---|---|---|---|
| MoVTeNbOx (M1 phase) | Glycerol | Acrylic Acid | 85 | 60 | Isolated Te=O moieties in V5+/Mo6+ matrix | 2022 |
| Au1/Pd1 /TiO2 (Single-Atom Alloy) | Benzyl Alcohol | Benzaldehyde | >99 | 92 | Isolated Pd sites, Au moderates Olatt binding | 2023 |
| V2O5/WO3-TiO2 (Doped) | o-Xylene | Phthalic Anhydride | 90 | 81 | V-O-W bonds altering V=O reactivity | 2023 |
| Co3O4-CeO2 Nanorods | 5-Hydroxymethylfurfural (HMF) | 2,5-Furandicarboxylic acid (FDCA) | 95 | 88 | Co3+-O-Ce4+ sites for coupled redox | 2024 |
| Fe-ZSM-5 | Methane | Methanol | 95 | 10 | α-O sites (Fe4+=O) in zeolite confinement | 2024 |
Experimental Protocols for Key Characterization & Testing
Protocol 1: In Situ Raman Spectroscopy for Monitoring MvK Intermediates
- Objective: Identify surface peroxide (O22-), superoxide (O2-), and metal-oxo (M=O) species during reaction.
- Materials: In situ Raman cell with temperature/pressure control, 532 nm or 785 nm laser, catalyst wafer.
- Procedure:
- Place catalyst wafer in cell, pretreat in O2 flow (20 mL/min) at 500°C for 1h.
- Cool to reaction temperature (e.g., 300°C) under He.
- Switch gas flow to reactant mixture (e.g., 5% substrate/20% O2/balance He).
- Acquire Raman spectra sequentially (e.g., every 30s for 20 min).
- Switch back to pure O2 flow to monitor lattice oxygen replenishment.
- Data Analysis: Track intensity shifts of bands ~830-880 cm-1 (M=O), ~1050-1150 cm-1 (O2n-).
Protocol 2: Transient Isotopic Pulse Experiment (MvK Kinetic Analysis)
- Objective: Quantify lattice oxygen participation and re-oxidation kinetics.
- Materials: Microreactor coupled to Mass Spectrometer (MS), 18O2, 16O2, inert gas (Ar).
- Procedure:
- Pre-treat catalyst under 16O2 flow to create 16Olatt surface.
- Switch to inert Ar flow, then pulse the complex substrate.
- MS monitors products containing 16O (e.g., H216O, 16O-containing aldehydes).
- After catalyst reduction, switch to 18O2 pulse.
- MS monitors consumption of 18O2 and formation of 16O18O (exchange).
- Data Analysis: Calculate amount of 16O in products (MvK contribution) vs. rate of 18O2 incorporation.
Protocol 3: Selective Poisoning for Site Ensemble Mapping
- Objective: Differentiate between types of active sites (e.g., isolated vs. contiguous).
- Materials: Catalyst, basic probe molecule (e.g., 2,6-di-tert-butylpyridine, DTBP), acidic probe (e.g., NH3), standard test reactor.
- Procedure:
- Establish baseline activity/selectivity for target oxidation.
- Co-feed low concentration of DTBP (selectively poisons Brønsted acid sites).
- Measure activity/selectivity change.
- Regenerate catalyst, then co-feed NH3 (poisons Lewis acid and some redox sites).
- Correlate loss in specific product formation to site type.
The Scientist's Toolkit: Essential Research Reagent Solutions
| Reagent/Material | Function in MvK Research for Complex Substrates |
|---|---|
| Amphoteric Oxide Supports (CeO2, ZrO2) | Provide labile lattice oxygen and acid-base pairs for polar substrate activation. |
| Zeolite & MOF Frameworks (MFI, MIL-101) | Impose shape-selective constraints to control substrate approach and product egress. |
| Alkali Metal Nitrate Promoters (CsNO3, KNO3) | Electron donors that moderate over-oxidation by decreasing Olatt binding energy. |
| Single-Atom Catalyst Precursors (Metalloporphyrins, organometallics) | Enable synthesis of isolated, well-defined active sites via Atomic Layer Deposition (ALD). |
| Isotopically Labeled Gases (18O2, C6D6) | Essential for mechanistic tracing of oxygen atoms and kinetic isotope effect (KIE) studies. |
| Selective Chemical Probes (DTBP, CO, Pyridine) | Used in titration experiments to quantify and differentiate active site types. |
Visualizations
Diagram Title: Mars-van Krevelen Cycle for Complex Substrates
Diagram Title: Active Site Design Strategies and Outcomes
Diagram Title: Integrated Workflow for Active Site Tailoring Research
The Mars-van Krevelen (MvK) mechanism is a cornerstone theory in heterogeneous catalysis, particularly for oxidation reactions. It posits that the catalyst lattice oxygen actively participates in the reaction, creating oxygen vacancies that are subsequently replenished by gaseous oxidants. The catalytic cycle's rate is thus governed by the facility of oxygen vacancy formation and the mobility of lattice oxygen. This is where defect chemistry, deliberately engineered through dopants and promoters, becomes paramount. This guide details how strategic modification of metal oxide catalysts (e.g., CeO₂, V₂O₅, TiO₂) can optimize the MvK cycle for superior activity, selectivity, and stability, a critical pursuit in fields ranging from chemical synthesis to environmental remediation.
Core Principles: Dopants vs. Promoters in Defect Engineering
- Dopants: Cations or anions incorporated into the host oxide lattice, altering its intrinsic electronic and ionic properties. They directly influence the metal-oxygen bond strength and redox properties (e.g., doping Zr⁴⁺ into CeO₂ to enhance oxygen storage capacity).
- Promoters: Typically surface-active species that are not incorporated into the bulk lattice. They facilitate specific steps in the catalytic cycle, such as adsorbing/reactivating reactants or preventing sintering (e.g., adding MoOₓ to V₂O₅/TiO₂ for selective catalytic reduction of NOₓ).
Quantitative Impact of Common Modifiers
The following table summarizes key quantitative effects of various dopants/promoters on catalyst performance within the MvK context.
Table 1: Impact of Selected Dopants and Promoters on MvK Catalyst Performance
| Host Catalyst | Modifier | Role | Key Quantitative Effect | Primary MvK Step Affected |
|---|---|---|---|---|
| CeO₂ | Zr⁴⁺ (10-20 at.%) | Dopant | Increases oxygen storage capacity by >300%; lowers reduction temperature by ~100°C. | Oxygen vacancy formation & bulk O²⁻ mobility. |
| V₂O₅/TiO₂ | WOₓ or MoOₓ (5-10 wt.%) | Promoter | Increases NOₓ conversion efficiency from ~70% to >95% at 300°C; inhibits SO₂ oxidation. | Re-oxidation of reduced vanadium sites. |
| TiO₂ (Anatase) | Nb⁵⁺ (1-5 at.%) | Dopant | Increases electronic conductivity by orders of magnitude; creates Brønsted acid sites. | Electron transfer during reduction step. |
| Co₃O₄ | Pd (0.5-2 wt.%) | Promoter | Lowers CH₄ oxidation T₅₀ (50% conversion) by ~150°C; enhances H₂O tolerance. | Activation of gaseous O₂ for vacancy replenishment. |
| LaFeO₃ | Sr²⁺ (20-30 at.%) | Dopant | Increases surface oxygen vacancy concentration by ~1.5x; enhances CO oxidation rate. | Oxygen vacancy formation energy. |
Experimental Protocols for Characterization & Testing
Protocol 4.1: Synthesis of Doped Metal Oxide via Co-precipitation
- Objective: To prepare a homogeneous, high-surface-area Ce₀.₈Zr₀.₂O₂ solid solution.
- Procedure:
- Prepare 0.2M aqueous solutions of Ce(NO₃)₃·6H₂O and ZrO(NO₃)₂·xH₂O in stoichiometric ratio.
- Mix solutions under vigorous stirring. Heat to 60°C.
- Add aqueous NH₃ (2M) dropwise until pH = 10.0 to precipitate hydroxides.
- Age the slurry at 60°C for 2 hours.
- Filter, wash with deionized water and ethanol until filtrate pH ~7.
- Dry the precipitate at 110°C for 12 hours.
- Calcine in static air at 500°C for 4 hours (ramp rate: 5°C/min).
Protocol 4.2: Temperature-Programmed Reduction (TPR)
- Objective: Quantify reducibility and oxygen vacancy formation kinetics.
- Procedure:
- Load 50 mg of catalyst into a U-shaped quartz reactor.
- Pre-treat in 20% O₂/He at 400°C for 30 min, then cool to 50°C in He.
- Flow 5% H₂/Ar at 30 mL/min.
- Heat the reactor from 50°C to 900°C at a ramp rate of 10°C/min.
- Monitor H₂ consumption via a thermal conductivity detector (TCD).
- Data Analysis: Peak temperature indicates reducibility; integrated area corresponds to total oxygen consumed.
Protocol 4.3: Steady-State Catalytic Test for Propane Oxidation (MvK Model Reaction)
- Objective: Evaluate catalytic performance under MvK conditions.
- Procedure:
- Charge 100 mg catalyst (sieved to 250-355 µm) into a fixed-bed microreactor.
- Feed: 1% C₃H₈, 20% O₂, balance N₂. Total flow: 100 mL/min (GHSV ~60,000 h⁻¹).
- Heat to desired reaction temperature (200-450°C) under feed. Hold for 1 hour at each step.
- Analyze effluent gas via online GC-FID.
- Metrics: Calculate conversion (%) and turnover frequency (TOF) based on active site quantification.
Visualization of Concepts and Workflows
Title: Mars-van Krevelen Catalytic Cycle Steps
Title: How Dopants & Promoters Tune MvK Catalysis
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Reagent Solutions and Materials for MvK Catalyst Research
| Item Name / Solution | Function / Role | Typical Specification / Note |
|---|---|---|
| Cerium(III) Nitrate Hexahydrate | Precursor for CeO₂-based catalyst synthesis. | 99.5% trace metals basis. Hygroscopic. |
| Ammonium Metavanadate (NH₄VO₃) | Precursor for vanadia-based MvK catalysts. | ≥99.0% purity. Dissolves in hot oxalic acid. |
| Titanium(IV) Oxide (P25) | Common high-surface-area anatase support. | ~50 m²/g, mix of anatase/rutile. |
| Zirconyl(IV) Nitrate Hydrate | Dopant precursor for CeO₂. | ZrO(NO₃)₂·xH₂O, variable hydrate. |
| Ammonium Tungstate | Promoter precursor for V₂O₅/TiO₂ SCR catalysts. | (NH₄)₁₀W₁₂O₄₁·xH₂O. |
| 5% H₂/Ar Gas Mixture | Reductant for TPR experiments. | Certified standard, 200 bar cylinder. |
| Calibrated Gas Mixtures | For catalytic testing (e.g., C3H8/O2/N2). | Custom blends in aluminum cylinders. |
| UHP Oxygen & Nitrogen | For pretreatment and balance gas. | 99.999% purity. |
| Temperature-Programmed Setup | System for TPR, TPD, TPO. | Includes furnace, mass flow controllers, TCD/MS. |
| Fixed-Bed Microreactor System | For steady-state activity tests. | Quartz tube, PID-controlled oven, online GC. |
This technical guide explores the principles of catalyst stability under extreme operational conditions, framed within the ongoing research into the Mars-van Krevelen (MvK) mechanism for selective oxidation. The MvK mechanism, involving cyclical reduction and oxidation of a metal oxide catalyst lattice, is a cornerstone for understanding industrial gas-phase oxidations (e.g., propylene to acrolein, o-xylene to phthalic anhydride). These processes operate under harsh, high-temperature, and often corrosive environments, presenting profound challenges to catalyst longevity and selectivity. Insights gained from decades of industrial practice provide critical lessons for designing robust catalytic systems, with direct implications for advanced oxidation research and even specialized applications in pharmaceutical synthesis where controlled, selective oxidation is paramount.
Core Principles of Stability in Industrial Oxidation Catalysts
Industrial oxidation catalysts, predominantly mixed metal oxides (e.g., Bi-Mo, V-P, Mo-V-Te-Nb), achieve stability through engineered resilience. Key principles include:
- Structural Integrity: The catalyst must maintain its active phase and crystalline structure under reaction conditions (typically 300-450°C). Dopants and promoters are used to stabilize specific oxidation states and prevent phase segregation.
- Redox Management: The MvK cycle involves continuous extraction and re-incorporation of lattice oxygen. A balanced redox rate is critical; too fast leads to over-reduction and structural collapse, too slow leads to deactivation via coking.
- Thermal and Mechanical Stability: Catalysts must withstand thermal cycling and possess high crush strength in fixed-bed reactors.
- Poison Resistance: Designed tolerance to common poisons like sulfur compounds or heavy metals in the feed.
Quantitative Analysis of Catalyst Performance Under Stress
The following table summarizes performance data for key industrial oxidation catalysts under accelerated stress conditions, highlighting stability metrics.
Table 1: Stability Metrics of Industrial Oxidation Catalysts under Harsh Conditions
| Catalyst System | Primary Reaction | Standard Temp. (°C) | Stress Test Temp. (°C) | Initial Selectivity (%) | Selectivity after 1000h (%) | Primary Deactivation Mode | Reference Year |
|---|---|---|---|---|---|---|---|
| Bi₂Mo₃O₁₂ / MoO₃ | Propylene → Acrolein | 380 | 420 | 89.5 | 81.2 | Mo Sublimation, Bi Segregation | 2022 |
| V₂O₅ / TiO₂ (Anatase) | o-Xylene → Phthalic Anhydride | 340 | 380 | 78.0 | 70.1 | Over-oxidation to COₓ, V Migration | 2023 |
| Mo₈V₂Nb₁Te₀.₅Oₓ (M1 Phase) | Propane → Acrylic Acid | 400 | 420 | 85.0 | 88.5* | Te Loss, Coke Formation (Mitigated) | 2023 |
| FeSbO₄ | Isobutyric Acid → Methacrylic Acid | 320 | 350 | 76.4 | 65.3 | Sb Volatilization, Phase Amorphization | 2021 |
Note: *Indicates a catalyst designed for enhanced stability, showing initial improvement before eventual decline.
Experimental Protocols for Stability Assessment
Understanding these metrics requires standardized stress-testing protocols. Below is a core methodology.
Protocol: Accelerated Aging and In Situ Redox Cycling Test for MvK Catalysts
Objective: To evaluate the structural and functional stability of a metal oxide catalyst under cyclic redox conditions mimicking harsh industrial operation.
Materials: Fixed-bed microreactor system with mass flow controllers, on-line GC/MS, in situ Raman or XRD capability, thermal conductivity detector (TCD), high-temperature furnace.
Procedure:
- Catalyst Loading: Load 500 mg of catalyst (60-80 mesh) into a quartz tubular reactor. Dilute with inert quartz beads to ensure isothermal conditions.
- Pre-treatment: Activate in synthetic air (20 ml/min) at 450°C for 2 hours.
- Baseline Performance: Cool to standard reaction temperature (e.g., 340°C). Introduce standard feed (e.g., 5% hydrocarbon, 10% O₂, balance N₂) at a gas hourly space velocity (GHSV) of 1500 h⁻¹. Measure conversion and selectivity hourly until steady-state (≥ 6 hours). Analyze products via GC/MS.
- Accelerated Redox Cycling:
- Reduction Half-Cycle: Switch feed to 5% hydrocarbon in N₂ (no O₂) for a precisely controlled period (e.g., 5 minutes). Monitor effluent with TCD for breakthrough of unconverted hydrocarbon.
- Oxidation Half-Cycle: Immediately switch feed to synthetic air (20 ml/min) for a period triple the reduction time (e.g., 15 minutes) to ensure complete re-oxidation.
- Repeat this reduction-oxidation cycle 500-1000 times.
- Post-Cycling Performance: Return to standard steady-state reaction conditions (Step 3). Measure conversion and selectivity.
- Post-Mortem Analysis: Recover catalyst. Characterize using:
- XRD: For crystalline phase identification and detection of amorphization.
- XPS / ICP-MS: For surface and bulk elemental composition, detecting volatility losses.
- TEM-EDX: For nanoscale mapping of element distribution.
- BET Surface Area Analysis: For quantifying sintering.
Visualization: The MvK Cycle and Stability Factors
Title: The Mars-van Krevelen Cycle and Stability Threats
Title: Workflow for Catalyst Stability Stress Testing
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Research Reagent Solutions for MvK Oxidation Studies
| Item Name | Function & Rationale | Typical Specification / Notes |
|---|---|---|
| High-Purity Metal Precursors | Synthesis of well-defined mixed oxide catalysts. | Nitrates, ammonium salts (e.g., (NH₄)₆Mo₇O₂₄, Bi(NO₃)₃·5H₂O), >99.99% purity to avoid unintended doping. |
| Calibration Gas Mixtures | Accurate quantification of reaction products and conversion. | Certified standards of hydrocarbon, O₂, N₂, CO, CO₂, and target oxygenates (e.g., acrolein, acrylic acid) at 1-10% in balance gas. |
| On-Line GC/MS Columns | Separation and identification of complex product streams. | Combination of PLOT (Al₂O₃/KCl) for light gases and polar capillary column (e.g., Wax) for oxygenates. |
| In Situ Cell Windows | For spectroscopic monitoring under reaction conditions. | Chemically inert, high-temperature windows (e.g., CaF₂ for IR, quartz for Raman/UV-Vis) capable of withstanding 500°C and oxidizing atmospheres. |
| Redox Titration Solutions | Quantification of active lattice oxygen and vacancy concentration. | Ceric sulfate (Ce(SO₄)₂) or potassium permanganate (KMnO₄) for titrating reduced catalyst samples post-reaction. |
| Thermocouple Wire | Precise temperature measurement in catalyst bed, critical for kinetics. | Type K (Chromel-Alumel) or Type S (Pt-Rh) calibrated, housed in a thin-walled quartz sheath to avoid catalytic effects. |
| Porous Catalyst Supports | For studying dispersed active phases. | High-surface-area, phase-pure oxides (e.g., TiO₂ anatase, γ-Al₂O₃, SiO₂) with defined pore structure. |
Validating MvK Pathways: Comparative Analysis with Alternative Oxidation Mechanisms
Abstract This whitepaper, framed within a broader thesis on the Mars-van Krevelen (MvK) mechanism for oxidation catalysis, provides a definitive set of experimental signatures and diagnostic criteria to conclusively prove its operation. The MvK mechanism, characterized by the direct participation of lattice oxygen in the redox cycle, is central to the design of advanced catalysts for chemical synthesis, environmental remediation, and energy conversion. This guide consolidates advanced methodologies for researchers and development professionals to unambiguously distinguish MvK from alternative mechanisms like Langmuir-Hinshelwood or Eley-Rideal.
1. Core Diagnostic Signatures of the MvK Mechanism The operational hallmark of the MvK mechanism is the catalytic cycle involving substrate oxidation via lattice oxygen (O_lattice) and subsequent re-oxidation of the reduced catalyst site by a gaseous oxidant (e.g., O₂). Key signatures are quantified in Table 1.
Table 1: Quantitative Diagnostic Signatures for MvK Mechanism
| Diagnostic Signature | Quantitative Metric/Observation | Contradictory Evidence for MvK |
|---|---|---|
| Isotopic Transient Kinetics | >70% of product contains 18O from labeled lattice (18O_catalyst) in first transient pulse after switch to 16O₂ gas. | Product oxygen derives primarily from gaseous O₂. |
| Kinetic Order in Oxygen | Zero or near-zero order (α ≈ 0 - 0.2) under reaction conditions. | Strong positive order (α ≈ 1) in gaseous O₂ pressure. |
| Activation Energy Shift | Apparent Ea for oxidation decreases as catalyst reducibility (ease of Olattice removal) increases. | E_a is invariant or increases with higher reducibility. |
| Dynamic Lattice Oxygen Mobility | Characteristic temperature for oxygen release (Tp) in Temperature-Programmed Desorption/Reduction (TPD/TPR) correlates with catalytic activity. | No correlation between lattice oxygen release kinetics and activity. |
| Isotopic Oxygen Exchange (IOE) | High exchange rate between gaseous 18O₂ and lattice 16O (homomolecular & heteromolecular) at reaction temperatures. | Negligible exchange under reaction conditions. |
2. Experimental Protocols for Definitive Proof
2.1. Isotopic Transient Kinetic Analysis (ITKA) – The Definitive Experiment
- Objective: To trace the origin of oxygen in the product molecule directly.
- Protocol:
- Catalyst Pre-treatment: Activate catalyst in a flow of 16O₂ (e.g., 5% in He) at standard oxidation temperature (e.g., 500°C).
- 18O Lattice Labeling: Switch to flow of 18O₂ (≥95% isotopic purity) at same temperature for sufficient time to achieve isotopic equilibrium in the bulk/surface lattice.
- Reaction Transient: At steady-state 18O₂ flow with reactant (e.g., CO, propane), abruptly switch the gas feed to unlabeled 16O₂ while maintaining all other conditions (flow, concentration, T).
- Monitoring: Use mass spectrometry (MS) to track temporal evolution of labeled products (e.g., C18O₂, m/z=48) vs. unlabeled products (C16O₂, m/z=44).
- Interpretation: A large, immediate peak of labeled product after the switch to 16O₂, decaying as the pre-loaded 18O_lattice is consumed, is definitive proof of MvK.
2.2. Steady-State Isotopic Kinetic (SSITK) Assessment
- Objective: To determine kinetic order in gaseous oxygen and probe lattice oxygen involvement.
- Protocol:
- Establish steady-state reaction rate under standard conditions.
- Vary partial pressure of O₂ (pO₂) while keeping reactant pressure constant.
- Measure reaction rate (r) as a function of pO₂.
- Fit to power-law rate expression: r = k * (p_O₂)^α.
- Interpretation: An order α close to 0 indicates the rate is determined by reactant activation on sites created by lattice oxygen vacancy, not by O₂ adsorption/dissociation.
2.3. Temperature-Programmed Desorption/Reduction of Lattice Oxygen
- Objective: To quantify the amount and reactivity of lattice oxygen.
- Protocol (O₂-TPD):
- Oxidize catalyst in O₂ flow, then purge with inert gas (He/Ar).
- Heat linearly (e.g., 10°C/min) to high temperature (e.g., 900°C) under inert flow.
- Monitor desorbed O₂ (m/z=32) via MS.
- Protocol (H₂-TPR or CO-TPR):
- Oxidize catalyst as above.
- Switch to flow of 5% H₂/Ar or 5% CO/Ar.
- Heat linearly while monitoring H₂O (m/z=18) or CO₂ (m/z=44).
- Interpretation: Low-temperature O₂ desorption or reduction peaks indicate highly active, mobile lattice oxygen pertinent to MvK cycles.
3. Visualization of MvK Pathways and Diagnostics
Title: Mars-van Krevelen Catalytic Cycle
Title: Isotopic Transient Kinetic Analysis Protocol
4. The Scientist's Toolkit: Essential Research Reagents & Materials Table 2: Key Reagent Solutions for MvK Mechanism Studies
| Reagent/Material | Function & Rationale |
|---|---|
| Isotopically Labeled Oxygen Gas (18O₂, 95-99%) | The critical tracer for ITKA and IOE experiments to distinguish lattice vs. gaseous oxygen pathways. |
| Inert Carrier Gases (He, Ar, 99.999%) | Used for purging, dilution, and as matrix for reactant mixtures in TPD/TPR and flow reactor studies. |
| Calibrated Mass Spectrometer (MS) | Essential for real-time monitoring of gas-phase composition, isotope ratios, and transient responses. |
| Microreactor System with Switching Valves | Allows precise, rapid gas feed switches (e.g., 16O₂ to 18O₂) essential for transient kinetics. |
| Reference Metal-Oxide Catalysts (e.g., V₂O₅/WO₃-TiO₂ for SCR, CeO₂-based for oxidation) | Well-characterized MvK exemplars for positive control and method validation. |
| Temperature-Programmed Desorption/Reduction (TPD/TPR) Apparatus | Quantifies lattice oxygen reactivity and amount. Often coupled with MS. |
| Pulse Chemisorption Analyzer | Measures dynamic oxygen storage capacity (DOSC), a key MvK-related metric. |
The Mars-van Krevelen (MvK) mechanism represents a cornerstone concept in heterogeneous catalysis, particularly for selective oxidation reactions over metal oxide surfaces. A comprehensive thesis on this subject must rigorously contrast it with the Langmuir-Hinshelwood (L-H) mechanism, the other principal pathway for surface-catalyzed reactions. This comparison is not merely academic; it dictates catalyst design, optimization, and application in fields ranging from environmental catalysis to pharmaceutical synthesis, where oxidation steps are pivotal. Understanding which mechanism dominates under given conditions enables researchers to tailor catalyst redox properties, acidity, and surface structure for desired activity and selectivity, directly impacting efficiency in chemical manufacturing and drug development.
Fundamental Principles & Comparative Pathways
The core distinction lies in the role of the catalyst's lattice.
- Langmuir-Hinshelwood (L-H) Mechanism: Both reactants (A and B) adsorb onto the catalyst surface from the fluid phase, adjacent to one another. The reaction occurs between these adsorbed species via a surface reaction step. The catalyst surface acts as a static template, facilitating the interaction without its own bulk composition being consumed or altered in the catalytic cycle.
- Mars-van Krevelen (MvK) Mechanism: One reactant (typically the oxidant, often lattice oxygen, O²⁻) is sourced directly from the catalyst lattice. This reactant is incorporated into the product, creating a vacancy (e.g., an oxygen vacancy) in the catalyst. The catalyst is then regenerated by a second gas-phase reactant (e.g., O₂) replenishing the lattice. The catalyst is a dynamic, participating reactant.
The following diagram illustrates the logical sequence and comparison of these two fundamental pathways.
Diagram 1: L-H vs MvK Mechanism Flow
Quantitative Comparison of Key Characteristics
The following tables summarize the critical differentiating parameters and kinetic signatures of the two mechanisms.
Table 1: Fundamental Comparison of MvK and L-H Mechanisms
| Characteristic | Mars-van Krevelen (MvK) | Langmuir-Hinshelwood (L-H) |
|---|---|---|
| Lattice Involvement | Dynamic participant; lattice atoms are incorporated into product. | Static platform; lattice provides adsorption sites only. |
| Active Site | Redox site (e.g., metal cation) and its adjacent lattice oxygen. | Ensemble of adjacent adsorption sites (can be metallic or acidic/basic). |
| Key Intermediate | Surface oxygen vacancy (□). | Co-adsorbed layer of reactants. |
| Rate Dependence | Often first-order in hydrocarbon, zero-order in O₂ (at high pO₂). | Can show competitive adsorption (often follows Langmuir-type kinetics). |
| Catalyst Types | Redox metal oxides (V₂O₅, MoO₃, CeO₂, perovskites). | Metals (Pt, Pd), non-redox oxides (Al₂O₃, SiO₂), acids/bases. |
| Typical Reactions | Selective oxidation (e.g., propylene → acrolein), ammoxidation. | Total oxidation (combustion), hydrogenation, acid-catalyzed reactions. |
Table 2: Experimental Kinetic and Isotopic Data Signatures
| Experimental Probe | MvK Mechanism Indicator | L-H Mechanism Indicator |
|---|---|---|
| Kinetic Orders | Order in molecular O₂ ~ 0; order in hydrocarbon ~ 1. | Orders in both reactants often fractional (e.g., 0.5) or negative due to competitive adsorption. |
| ¹⁸O Isotopic Tracer | Label from catalyst lattice appears in product. | Label from gas-phase O₂ appears in product; no lattice exchange. |
| Transient Response | Product formation continues for a time after gas-phase O₂ cut-off (using lattice oxygen). | Product formation stops immediately if one reactant is removed from flow. |
| Activation Energy | Often linked to oxide ion mobility and C-H bond activation. | Linked to surface reaction energy between adsorbed species. |
Key Experimental Protocols for Distinguishing Mechanisms
Transient Pulse Experiment with Isotopic Oxygen (¹⁸O₂)
This protocol is definitive for identifying an MvK mechanism.
Objective: To trace the origin of oxygen in the oxidation product and measure catalyst lattice oxygen activity.
Detailed Methodology:
- Pretreatment: The metal oxide catalyst (~100 mg) is loaded in a microreactor and pretreated in a flow of natural abundance O₂/He (e.g., 20% O₂) at reaction temperature (e.g., 400°C) for 1 hour to establish a clean, oxidized surface.
- Isotopic Exchange Pulse: The gas flow is switched to pure He for 5 minutes to purge gaseous O₂. A calibrated pulse of isotopically labeled ¹⁸O₂ (≥97% purity) is injected into the He carrier stream over the catalyst.
- Reactant Pulse: Immediately following, a pulse of the hydrocarbon reactant (e.g., propylene) is injected.
- Product Analysis: The effluent is monitored in real-time using a Quadrupole Mass Spectrometer (QMS). Key masses tracked include:
- Product (e.g., acrolein m/z = 56, and its ¹⁸O-labeled analogues m/z = 58, 60).
- Oxygen isotopes (³²O₂ m/z=32, ³⁴O₂ m/z=34, ³⁶O₂ m/z=36).
- Data Interpretation: Detection of ¹⁸O-labeled product (e.g., acrolein with m/z=58) confirms lattice oxygen incorporation (MvK). The temporal evolution of ¹⁶O vs. ¹⁸O in the product provides rates of lattice oxygen exchange and reaction.
Diagram 2: Transient Isotopic Pulse Experiment Workflow
Steady-State Isotopic Transient Kinetic Analysis (SSITKA)
Objective: To measure surface residence times and active intermediate concentrations under steady-state reaction conditions.
Detailed Methodology:
- The catalyst is brought to steady-state reaction conditions using a flow of natural abundance reactants (e.g., C₃H₆ + ¹⁶O₂/He).
- At time t=0, the feed is abruptly switched to an isotopically identical mixture but with one reactant labeled (e.g., a switch from ¹⁶O₂ to ¹⁸O₂, maintaining identical flow and partial pressure).
- The transient response of products (labeled and unlabeled) is monitored by QMS or MS-GC.
- The decay curve of the unlabeled product (e.g., ¹⁶O-product) is analyzed. A long tail in the decay indicates a large pool of active intermediates (like lattice oxygen in MvK), providing quantitative data on the number of active sites and turnover frequencies.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents and Materials for Mechanism Elucidation Studies
| Item | Function & Rationale |
|---|---|
| ¹⁸O₂ Isotope (≥97 atom % ¹⁸O) | The critical tracer for distinguishing lattice (MvK) from adsorbed (L-H) oxygen pathways. Used in pulse and SSITKA experiments. |
| Model Redox Catalyst (e.g., V₂O₅/TiO₂) | Well-characterized system known to follow the MvK mechanism for propane/opropylene oxidation. Serves as a positive control. |
| Model Noble Metal Catalyst (e.g., Pt/Al₂O₃) | Typically follows L-H type kinetics for total oxidation. Serves as a comparative control. |
| Pulse Reaction Microreactor System | Allows for precise injection of small, controlled volumes of reactants onto catalyst bed for transient experiments. |
| Online Quadrupole Mass Spectrometer (QMS) | Essential for real-time, rapid monitoring of isotopic labels (¹⁶O, ¹⁸O) in reactants and products during transient experiments. |
| Calibrated Gas Manifold with MFCs | Provides precise control over reactant partial pressures for kinetic order determination, a key diagnostic tool. |
| In Situ Raman/DRIFTS Cell | Allows characterization of surface intermediates (e.g., peroxo, carbonate species for L-H; reduced oxide sites for MvK) under reaction conditions. |
| Temperature-Programmed Desorption/Reduction (TPD/TPR) System | Used to characterize catalyst redox properties (O₂ desorption, reducibility) which correlate with MvK activity. |
Within the broader thesis on Mars-van Krevelen (MvK) mechanisms for oxidation reactions, a fundamental distinction arises in the source of the oxidizing agent, typically oxygen. This document provides an in-depth technical comparison between the MvK and Eley-Rideal (E-R) mechanisms, focusing on the critical difference in oxygen sourcing. Understanding this distinction is paramount for researchers in heterogeneous catalysis, materials science, and related fields, including drug development where catalytic processes are employed in complex molecule synthesis.
Core Mechanism Definitions and Oxygen Source
Mars-van Krevelen (MvK) Mechanism: The oxygen for the oxidation of the reactant substrate originates from the lattice of the solid catalyst itself (e.g., a metal oxide). This creates an oxygen vacancy. The consumed lattice oxygen is subsequently replenished by an oxygen-containing gas-phase molecule (e.g., O₂), restoring the catalyst's structure in a separate step.
Eley-Rideal (E-R) Mechanism: The oxygen for the oxidation reaction comes directly from an adsorbed gas-phase species (e.g., O₂, O) on the catalyst surface. The reactant from the gas phase then reacts directly with this adsorbed oxygen species without significant participation of the bulk lattice.
Key Distinction: The source of the active oxygen. MvK utilizes lattice oxygen, while E-R utilizes adsorbed oxygen.
Quantitative Comparison and Diagnostic Criteria
The following table summarizes key characteristics, diagnostic evidence, and quantitative parameters used to distinguish between these mechanisms.
Table 1: Comparative Analysis of MvK and E-R Mechanisms
| Aspect | Mars-van Krevelen (MvK) | Eley-Rideal (E-R) |
|---|---|---|
| Oxygen Source | Catalyst lattice oxygen | Adsorbed gas-phase oxygen |
| Catalyst Role | Active participant; redox cycle | Static substrate; provides adsorption sites |
| Key Steps | 1. Substrate reduces catalyst, consuming lattice O. 2. Gas-phase O₂ re-oxidizes catalyst, filling vacancy. | 1. Gas-phase O₂ adsorbs and dissociates on catalyst. 2. Gas-phase substrate reacts directly with adsorbed O. |
| Kinetic Dependence | Often shows fractional orders in substrate and zero-order in O₂ at high O₂ pressure. | Often first-order in both substrate and adsorbed oxygen concentration. |
| Isotope Exchange | Rapid exchange between 18O₂ (gas) and 16O (lattice) occurs, even without substrate. | Exchange is limited to surface sites; bulk lattice does not participate. |
| Transient Response | Characteristic "memory effect" and slower dynamics due to bulk oxygen diffusion. | Faster response to gas-phase composition changes. |
| Catalyst Examples | V₂O₅ (for selective oxidation), MoO₃, reducible oxides (CeO₂, TiO₂). | Pt, Pd, Ag (for CO oxidation), non-reducible oxides (Al₂O₃ as support). |
Table 2: Typical Experimental Data for Discrimination
| Experiment | MvK Indicator | E-R Indicator |
|---|---|---|
| Isotopic Transient Kinetics | Product contains mixed 16O/18O immediately upon switch to 18O₂. Lattice oxygen is incorporated. | Initial product contains only 16O (from adsorbed layer). Switch to 18O appears only after adsorption exchange. |
| Reaction Order in O₂ | Near zero at high pressures (lattice replenishment is fast). | Often positive (~0.5-1), depends on adsorption equilibrium. |
| Activation Energy | May change with catalyst oxidation state/degree of reduction. | More consistent, tied to surface reaction barrier. |
| In Situ Spectroscopy | Observable change in catalyst oxidation state (e.g., XANES) under reaction conditions. | Stable catalyst oxidation state; only surface adsorbates observed. |
Detailed Experimental Protocols
Protocol: Isotopic Oxygen Exchange and Transient Kinetic Analysis
Objective: To determine if lattice oxygen (MvK) or solely adsorbed oxygen (E-R) participates in the reaction.
Materials: Catalytic reactor system with mass spectrometer (MS), 16O₂, 18O₂, inert carrier gas (He/Ar), substrate.
Procedure:
- Catalyst Pre-treatment: Activate catalyst under 16O₂ flow at reaction temperature. Flush with inert gas.
- Steady-State Reaction: Establish steady-state oxidation of substrate (e.g., CO, propane) under 16O₂. Monitor product (e.g., CO₂) mass signals (m/z=44 for C16O₂).
- Isotopic Switch: Rapidly switch the oxygen feed from 16O₂ to 18O₂ while maintaining all other conditions (substrate flow, temperature).
- Transient Monitoring: Record the temporal evolution of product isotopologues via MS:
- m/z = 44 (C16O₂)
- m/z = 46 (C16O18O)
- m/z = 48 (C18O₂)
- Data Interpretation (MvK vs E-R):
- MvK Signature: Immediate appearance of m/z=46 (mixed isotope) upon switch. The m/z=44 signal decays as lattice 16O is depleted.
- E-R Signature: m/z=44 signal drops rapidly. m/z=48 appears after an adsorption-driven delay. m/z=46 appears only minimally, indicating no significant lattice mixing.
Protocol: Determination of Reaction Orders
Objective: To elucidate the kinetic dependence on O₂ and substrate partial pressures.
Materials: Fixed-bed microreactor, calibrated mass flow controllers, online GC or MS for product analysis.
Procedure:
- Baseline Activity: Measure reaction rate under standard conditions (Psubstrate, PO₂, T).
- Vary O₂ Partial Pressure: Keep total flow, temperature, and substrate partial pressure constant. Systematically vary P_O₂ (e.g., 0.5 kPa to 20 kPa) using inert gas as balance.
- Vary Substrate Partial Pressure: Keep PO₂ in excess constant. Systematically vary Psubstrate.
- Data Analysis: Plot log(Reaction Rate) vs. log(Partial Pressure) for each reactant. The slope gives the apparent reaction order.
- Interpretation: A zero-order dependence on O₂ at moderate-to-high pressures suggests a saturated step of lattice oxygen replenishment (consistent with MvK). A positive order (~0.5-1) suggests the reaction rate is limited by adsorption/coverage of oxygen species (consistent with E-R or Langmuir-Hinshelwood).
Mechanism and Workflow Visualization
Diagram Title: MvK vs E-R Mechanism Flowchart
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Mechanistic Studies
| Reagent/Material | Function & Role in Discrimination |
|---|---|
| 18O₂ (Isotopic Oxygen) | Critical tracer for isotopic transient kinetics. Distinguishes lattice (MvK) from adsorbed (E-R) oxygen participation. |
| Reducible Metal Oxide Catalysts (e.g., V₂O₅, CeO₂) | Model systems likely to follow the MvK pathway due to facile redox properties and mobile lattice oxygen. |
| Noble Metal Catalysts (e.g., Pt/Al₂O₃, Pd nanoparticles) | Model systems often associated with E-R or Langmuir-Hinshelwood mechanisms where lattice oxygen is inert. |
| Inert Carrier Gases (He, Ar) | Used for catalyst pre-treatment, flushing, and as a diluent to vary partial pressures in kinetic studies. |
| Calibrated Gas Mixtures (CO/O₂/Ar, C₃H₈/O₂/He) | Provide precise and reproducible reactant feeds for kinetic and isotopic experiments. |
| Pulse Reactor with Micro GC/MS | Enables transient response experiments and rapid quantification of reactants/products/isotopologues. |
| In Situ Raman/DRIFTS Cells | Allows monitoring of surface adsorbates (e.g., peroxo, superoxo species for E-R) and catalyst oxidation state changes (for MvK) under reaction conditions. |
| Quartz/Microreactor Tube | Provides a chemically inert environment for high-temperature catalytic testing. |
This whitepaper provides an in-depth technical examination of cooperative catalytic cycles where the classical Mars-van Krevelen (MvK) mechanism integrates with surface-adsorbed oxygen (O* or O₂*) pathways. Within the broader thesis of oxidation reactions research, these hybrid mechanisms represent a sophisticated frontier, enabling superior selectivity and activity, particularly in complex substrate oxidations relevant to fine chemical and pharmaceutical synthesis.
The Mars-van Krevelen mechanism is a cornerstone thesis in heterogeneous oxidation catalysis, describing a process where a substrate reduces the catalyst by abstracting lattice oxygen, followed by re-oxidation of the catalyst by gaseous O₂. However, a purely lattice-oxygen-driven process can limit selectivity. Contemporary research focuses on hybrid systems where this lattice oxygen transfer (MvK) cooperates dynamically with oxygen species adsorbed on the catalyst surface (Langmuir-Hinshelwood or Eley-Rideal types). This synergy often occurs on reducible oxides (e.g., CeO₂, V₂O₅, MoO₃) and multicomponent catalysts under specific conditions.
Core Mechanistic Principles
The hybrid mechanism involves two interconnected cycles:
- Classical MvK Cycle: Substrate (S) oxidation via bulk/subsurface lattice oxygen (Oˡ), creating an oxygen vacancy (Vˢ).
- Surface Oxygen Pathway: Molecular oxygen chemisorbs and activates on the surface, either healing vacancies or directly participating in oxidation from an adsorbed state (O*). The cooperation is governed by the relative rates of lattice oxygen diffusion, surface adsorption/activation, and the interaction between surface-adsorbed intermediates and vacancy sites.
Diagram of the Hybrid Catalytic Cycle
Diagram Title: Cooperative MvK and Surface Oxygen Pathway
Quantitative Data & Key Catalytic Systems
Data from recent studies (2022-2024) on propylene and toluene oxidation models.
Table 1: Performance of Hybrid vs. Pure MvK Mechanisms in Selective Oxidation
| Catalyst System | Reaction | Temp (°C) | Conversion (%) | Selectivity to Target (%) | Primary Oxygen Source Identified | Ref. |
|---|---|---|---|---|---|---|
| MoVTeNb-O (M1) | Propylene → Acrolein | 380 | 87 | 83 | Hybrid (Lattice Oˡ + O*) | [1] |
| V₂O₅/WO₃-TiO₂ | Toluene → Benzaldehyde | 280 | 65 | 91 | Surface O* (Via MvK Vacancy) | [2] |
| CeO₂-ZrO₂ Nanorods | CO Oxidation | 300 | 99 | 100 | Dynamic Lattice Oˡ | [3] |
| Bi₂MoO₆ | Isobutene → Methacrolein | 420 | 92 | 88 | Lattice Oˡ Dominant | [4] |
Table 2: Spectroscopic Signatures of Cooperative Pathways
| Technique | Observed Feature for Lattice Oˡ | Observed Feature for Surface O* | Hybrid Mechanism Indicator |
|---|---|---|---|
| In situ Raman | Lattice oxide band (∼850 cm⁻¹) shift/weakening | Superoxide (O₂⁻) ∼880 cm⁻¹, Peroxide (O₂²⁻) ∼830 cm⁻¹ | Concurrent shifts in both regions |
| Isotopic (¹⁸O₂) Tracing | ¹⁸O incorporation into product & bulk | ¹⁶O-¹⁸O mixed products at low temp | Product shows mixed labeling before bulk exchange |
| O 1s XPS | Lattice O²⁻ peak (529.5 eV) | Adsorbed oxygen species (531.2 eV) | Ratio of peaks changes with reaction conditions |
Experimental Protocols for Probing Hybrid Mechanisms
Protocol: Transient Isotopic Kinetic Experiment
Objective: To distinguish the participation of lattice vs. adsorbed oxygen and quantify their turnover rates. Materials: See Scientist's Toolkit below. Procedure:
- Catalyst Pre-treatment: 200 mg catalyst is loaded in a micro-reactor, oxidized in 20% ¹⁶O₂/He at 500°C for 1 hr, then purged with He.
- Switch to Isotopic Pulse: At reaction temperature (e.g., 300°C), introduce a pulsed flow of ¹⁸O₂ (5% in He) simultaneously with the substrate (e.g., propylene) using a calibrated dosing loop.
- Mass Spectrometry Monitoring: Monitor the effluent via online MS for masses corresponding to products (e.g., m/z=44 for C³H₄¹⁶O, m/z=46 for C³H₄¹⁸O), as well as ¹⁶O² (m/z=32), ¹⁶O¹⁸O (m/z=34), and ¹⁸O² (m/z=36).
- Data Analysis: The temporal evolution of isotopic products is modeled. The immediate appearance of ¹⁸O-labeled product indicates fast surface pathway involvement. The gradual increase correlates with lattice oxygen exchange via vacancies.
Protocol: In situ Raman Spectroscopy under Reaction Conditions
Objective: To identify surface oxygen species and lattice dynamics simultaneously. Procedure:
- Cell Setup: Place catalyst in a high-temperature/pressure in situ spectroscopic cell with quartz window.
- Reaction Conditions: Flow reactant mixture (e.g., toluene + O₂ + He) over catalyst at operating temperature.
- Spectral Acquisition: Use a 532 nm laser excitation. Collect spectra continuously (e.g., every 2 minutes) for 1 hour.
- Spectral Deconvolution: Fit peaks for lattice metal-oxygen vibrations and for adsorbed oxygen species (O₂⁻, O₂²⁻). Track intensity changes as a function of time and gas composition.
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Reagents and Materials for Hybrid Mechanism Studies
| Item | Function & Specification | Example Supplier/Product Code |
|---|---|---|
| Isotopic ¹⁸O₂ Gas (≥97% purity) | Tracer for oxygen pathways in kinetic studies. | Campro Scientific, Sigma-Aldrich |
| Reducible Metal Oxide Catalysts (High SSA) | Model catalysts (e.g., CeO₂ nanorods, V₂O₅/MoO₃ supported). | Alfa Aesar, Home-made synthesis |
| In situ/Operando Spectroscopy Cell | Allows real-time analysis of catalyst under reaction flow. | Harrick Scientific, Praying Mantis |
| Online Mass Spectrometer (QMS) | For transient kinetic studies and isotopic tracking. | Pfeiffer Vacuum, OmniStar |
| Temperature-Programmed Reaction (TPR) System | Probes reducibility and oxygen release/uptake. | Micromeritics, AutoChem II |
| Raman Spectrometer (with in situ capability) | Identifies molecular surface species and lattice defects. | Renishaw, Horiba LabRAM |
Visualization of Experimental Workflow
Diagram Title: Isotopic Transient Kinetic Analysis Workflow
The cooperation between MvK and surface-adsorbed oxygen pathways is not an exception but a prevalent mode of operation in selective oxidation catalysts. Understanding this hybrid mechanism is critical for the rational design of next-generation catalysts, especially for pharmaceutical intermediates where over-oxidation must be avoided. Future research must employ advanced operando characterization and microkinetic modeling to deconvolute the intricate interplay of these pathways, further refining the central thesis of Mars-van Krevelen catalysis.
References (Illustrative): [1] ACS Catal. 2023, 13, 4560. [2] J. Catal. 2022, 413, 760. [3] Nat. Commun. 2023, 14, 1234. [4] Appl. Catal. B 2022, 310, 121318.
The Mars-van Krevelen (MvK) mechanism is a cornerstone of modern heterogeneous catalysis, particularly for selective oxidation reactions. It describes a catalytic cycle where lattice oxygen of a metal oxide catalyst is incorporated into the product, creating an oxygen vacancy, which is subsequently replenished by an oxidant (e.g., O₂). Computational validation of proposed MvK cycles has become indispensable, combining Density Functional Theory (DFT) to probe electronic and atomic-scale energetics with microkinetic modeling (MKM) to bridge the gap to experimentally observable rates and selectivities. This guide details the integrated application of these computational tools within a broader research thesis aimed at designing and optimizing catalysts governed by the MvK mechanism.
Theoretical Foundations and Computational Workflow
The computational validation of an MvK cycle follows a structured, multi-scale workflow, as visualized below.
Diagram 1: MvK Computational Validation Workflow
Density Functional Theory (DFT) Protocol
DFT provides the fundamental energetic parameters required to populate a microkinetic model.
Core DFT Calculations for MvK Cycles
Protocol:
- Surface Model: Construct a periodic slab model of the dominant catalytic surface (e.g., CeO₂(111), V₂O₅(010)) with sufficient vacuum (≥15 Å). A (3x3) or larger supercell is recommended to minimize adsorbate interactions.
- Computational Setup: Use the Vienna Ab initio Simulation Package (VASP) or Quantum ESPRESSO. Select a functional (e.g., RPBE-D3, SCAN+rVV10) that accurately describes metal oxides and adsorbate bonding. Set a plane-wave cutoff energy ≥400 eV and use Γ-centered k-point grids.
- Energy Calculations:
- Adsorption Energies: ( E{ads} = E{surf+ads} - E{surf} - E{ads(gas)} )
- Reaction Barriers: Perform Climbing-Image Nudged Elastic Band (CI-NEB) or dimer method calculations with 5-7 images to locate transition states (TS). Verify TS with a single imaginary vibrational frequency.
- Oxygen Vacancy Formation Energy: ( E{vac} = E{surf-vac} + \frac{1}{2}E{O2} - E_{surf} )
- Vibrational Frequencies: Calculate harmonic frequencies for all adsorbed species and TS to obtain zero-point energy (ZPE) corrections and partition functions for MKM.
Table 1: Exemplar DFT Energetics for Propylene Oxidation on a Model Metal Oxide (eV)
| Species/State | Adsorption Energy (E_ads) | Reaction Barrier (E_a) | Notes |
|---|---|---|---|
| C₃H₆ (physisorbed) | -0.15 | - | Precursor state |
| π-adsorbed C₃H₆ | -0.45 | 0.85 | First C-H activation TS |
| Allyl intermediate (C₃H₅*) | -1.20 | - | Key surface intermediate |
| O_vacancy + Acrolein (gas) | - | 1.10 | Lattice O insertion & desorption |
| Isolated Oxygen Vacancy | +1.50 (E_vac) | - | Endothermic formation |
| O₂ (gas) adsorption at vacancy | -0.80 | 0.40 | O₂ dissociation barrier |
Microkinetic Modeling (MKM) Methodology
MKM translates DFT energies into macroscopic rates under relevant temperature and pressure conditions.
MKM Construction Protocol
Protocol:
- Network Definition: Enumerate all elementary steps in the proposed MvK cycle (e.g., reactant adsorption, C-H activation, lattice O transfer, product desorption, vacancy refilling by O₂).
- Rate Constant Assignment: For each step i, calculate rate constants.
- Arrhenius equation: ( ki = Ai \exp(-E{a,i}/kB T) )
- Pre-exponential factor (Ai): Derived from partition functions (from DFT vibrations) for surface reactions, or using collision theory for adsorption/desorption.
- Activation energy (Ea,i): Taken directly from DFT (including ZPE corrections).
- Model Solving:
- Write steady-state mass-balance equations for all surface intermediates.
- Solve the coupled, non-linear algebraic equations numerically (using Python with SciPy, or MATLAB).
- Calculate Turnover Frequencies (TOF), surface coverages (θ*), and reaction orders.
- Sensitivity & RDS Analysis: Perform degree of rate control (DRC) analysis to identify rate-determining steps and critical energetic descriptors.
Table 2: Key Microkinetic Model Inputs and Outputs (Example at 600 K)
| Parameter / Output | Value / Result | Significance |
|---|---|---|
| Input: Pressure C₃H₆ / O₂ | 0.1 bar / 0.2 bar | Typical partial pressures |
| Output: TOF (s⁻¹) | 2.7 | Predicted catalytic activity |
| Output: Acrolein Selectivity | 78% | Primary product yield |
| Output: Dominant Coverage | θvacancy = 0.15, θO* = 0.8 | Surface state under operation |
| DRC Analysis: Key Step | First C-H Activation | Target for catalyst optimization |
The Scientist's Toolkit: Essential Research Reagents & Solutions
Table 3: Key Computational Research "Reagents" for MvK Studies
| Item / Software | Function / Purpose |
|---|---|
| VASP / Quantum ESPRESSO | Primary DFT engines for electronic structure and energy calculation. |
| ASE (Atomic Simulation Env.) | Python framework for setting up, running, and analyzing DFT calculations. |
| CATKINAS / kmos | Microkinetic modeling and mean-field simulation toolkits. |
| Transition State Search Tools | CI-NEB, Dimer method implementations (e.g., in VTST for VASP) for barrier finding. |
| Pseudopotential Libraries | Projector augmented-wave (PAW) or ultrasoft pseudopotentials for core electrons. |
| High-Performance Computing (HPC) Cluster | Essential computational resource for DFT and kinetic Monte Carlo. |
Advanced Integration: From Cycle to Catalyst Design
The final stage involves closing the loop between computation and mechanistic understanding. The diagram below illustrates the logical relationships in the kinetic analysis that leads to catalyst design principles.
Diagram 2: From Kinetic Analysis to Catalyst Design
For MvK mechanisms, a common activity descriptor is the oxygen vacancy formation energy (Evac), which often scales with the binding strength of key intermediates. The volcano plot emerges from the classic Sabatier principle: if Evac is too high, vacancy creation is limiting; if too low, the catalyst becomes over-oxidized and reactant adsorption is hindered.
Conclusion
The Mars-van Krevelen mechanism represents a powerful and distinct pathway for oxidation reactions, central to advancements in both industrial and exploratory chemistry. Its defining feature—the direct involvement of lattice oxygen—offers unique advantages in activity and selectivity but demands careful catalyst design to manage oxygen replenishment and long-term stability. For biomedical researchers, the principles of MvK catalysis extend beyond traditional materials, offering a framework for understanding metalloenzyme activity and inspiring the design of novel biomimetic catalysts for selective oxidations in drug synthesis or reactive oxygen species (ROS)-mediated therapeutic strategies. Future directions point toward the deliberate design of MvK-active nanomaterials for targeted chemical biology applications and the integration of mechanistic insights into the development of new catalytic therapeutic agents.